Introduction
Understanding the complexities of hazardous pharmaceutical waste disposal is essential for healthcare facilities. Mismanagement can lead to severe consequences for public health and the environment. This article explores best practices that ensure compliance with stringent regulations while safeguarding communities from the risks associated with improper disposal. Given the alarming statistics regarding the impact of pharmaceutical waste on health and ecosystems, healthcare providers must navigate the challenges of safe disposal while adhering to legal standards.
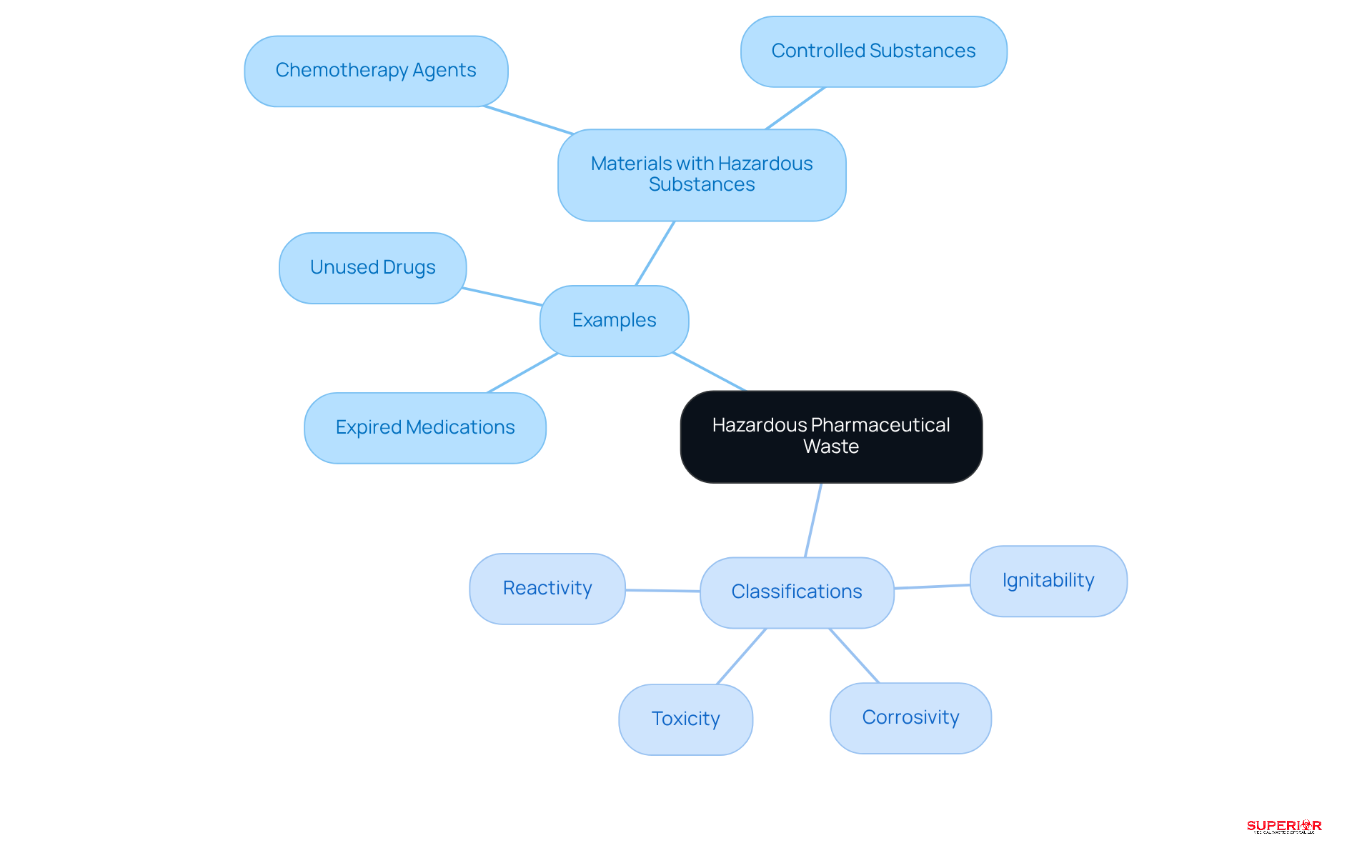
Define Hazardous Pharmaceutical Waste
refers to any pharmaceutical item deemed dangerous due to its potential to harm human health or the environment. This category includes:
- Unused drugs
- Materials containing hazardous substances, such as chemotherapy agents and
The Environmental Protection Agency (EPA) classifies these based on specific characteristics:
- Ignitability
- Corrosivity
- Reactivity
- Toxicity
For example, substances that can easily ignite or are corrosive to human tissue fall under these classifications.
Understanding these classifications is crucial for healthcare establishments to ensure . Mismanagement can lead to severe and environmental damage. In fact, improper disposal practices contribute to an estimated 5.2 million deaths annually worldwide due to unmanaged medical refuse, underscoring the urgent need for .
Moreover, all establishments producing medical refuse are legally required to have their sharps and removed from their premises every 90 days or less. They must also obtain a Certificate of Registration, which expires every three years and incurs varying fees based on the type of establishment. As emphasized by , adhering to these classifications and regulations not only protects public health but also ensures compliance with legal standards, allowing healthcare establishments to operate within legal frameworks while safeguarding the environment.
Understand Regulatory Requirements for Disposal
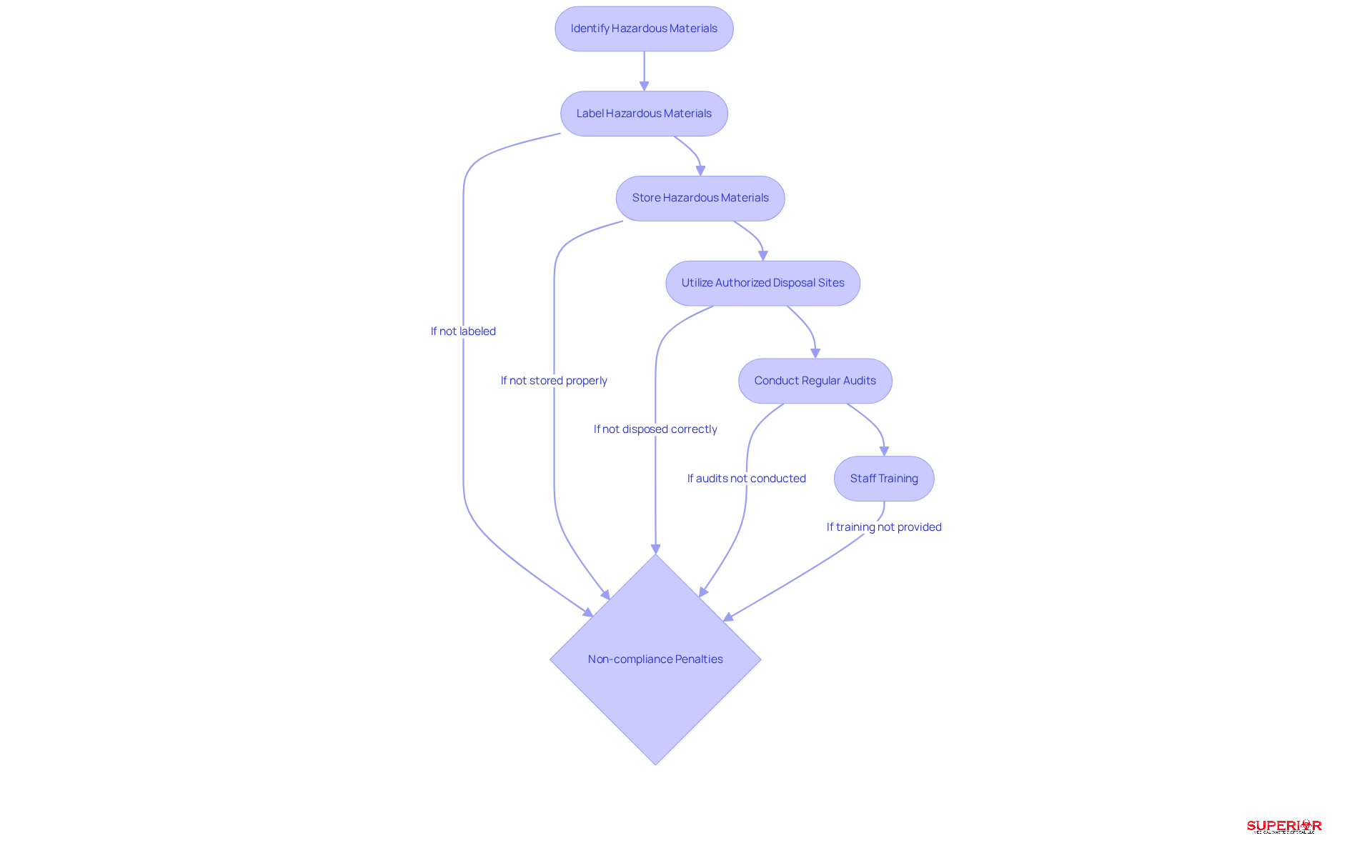
Healthcare establishments must comply with stringent regulatory standards when managing , primarily governed by the . This legislation mandates that these establishments accurately identify, label, and store hazardous materials in accordance with .
The has outlined specific guidelines for hazardous pharmaceutical waste disposal, emphasizing the necessity of utilizing authorized sites for incineration or safe landfilling. Regular audits and comprehensive are crucial for maintaining , ensuring that all personnel are well-informed about their roles and responsibilities in resource management.
Statistics underscore the importance of adhering to , as non-compliance can result in . By implementing these best practices, healthcare facilities can significantly mitigate risks associated with hazardous pharmaceutical waste disposal, thereby upholding safety and environmental integrity.
Identify Risks of Improper Disposal
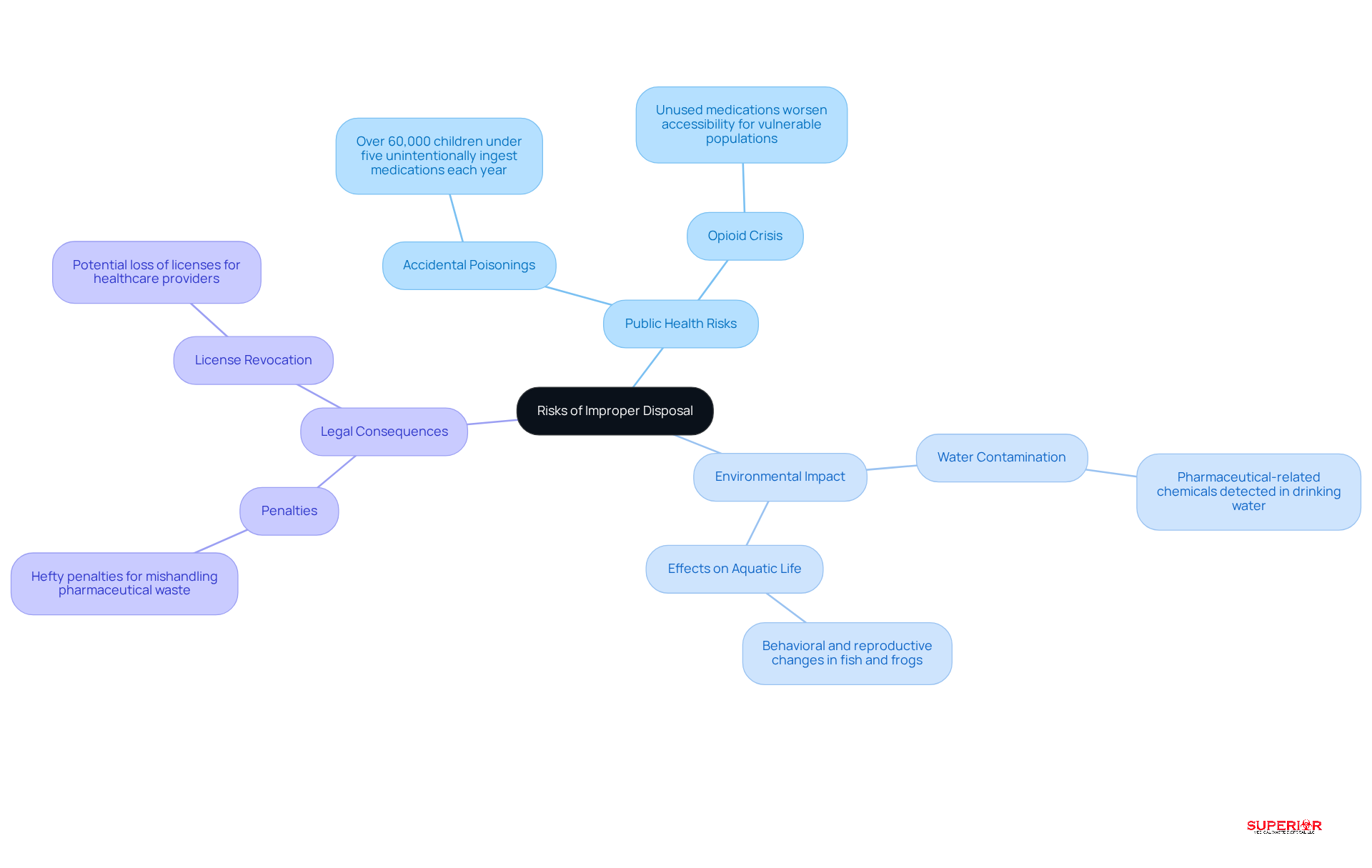
The presents significant risks to public health and the environment. When pharmaceuticals enter the waste stream, they can , leading to ecological damage and posing health risks to communities. Research has shown that pharmaceutical-related chemicals have been detected in drinking water, raising concerns about their effects on human health and aquatic life. For example, inadequate waste management practices can result in drugs leaching into waterways, which has been linked to behavioral and reproductive changes in species such as fish and frogs.
Additionally, the risk of accidental poisonings increases, particularly among children who may confuse medications with candy. Statistics reveal that over 60,000 children under five unintentionally ingest medications each year, highlighting the . Furthermore, the availability of unused medications can , as these substances remain accessible to vulnerable populations.
for mishandling pharmaceutical waste, including hefty penalties and potential revocation of licenses. This reality underscores the importance of recognizing these risks as a driving force for healthcare providers to adopt . By implementing organized removal methods, such as drug take-back initiatives and utilizing comprehensive provided by Superior Medical Waste Management – including and digital shipping documentation – healthcare establishments can mitigate these risks and contribute to a safer environment.
Implement Safe Disposal Strategies
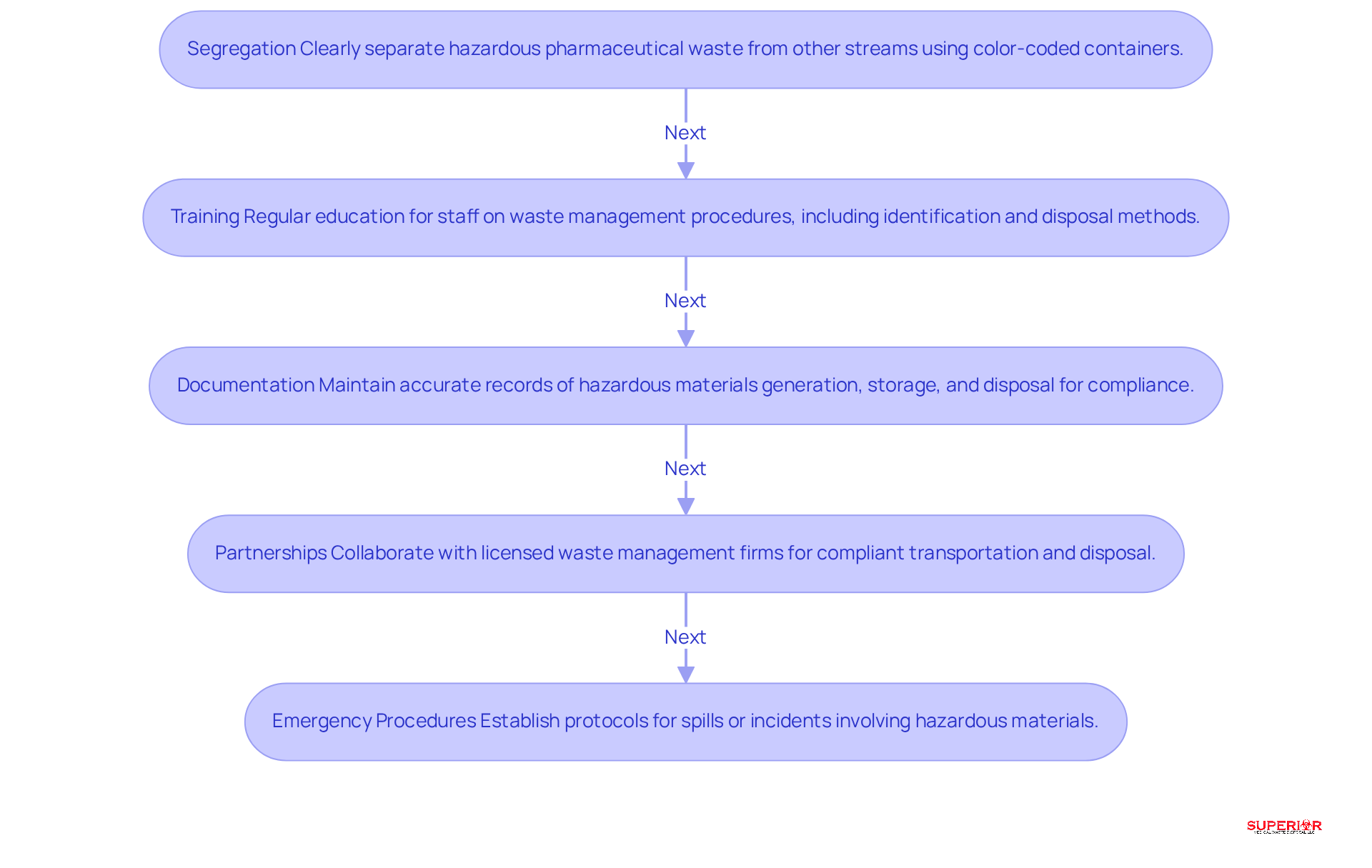
To implement safe disposal strategies for , healthcare facilities should adopt several best practices:
- Segregation: It is crucial to clearly separate hazardous pharmaceutical waste from other disposal streams to prevent contamination. Utilizing specifically designed for hazardous materials significantly enhances sorting compliance.
- Training: Regular education for staff on -including identification, segregation, and disposal methods-is essential. offers extensive , which includes specific modules on handling pharmaceutical materials. This ensures that staff remains informed about legal requirements and best practices. Research indicates that educating healthcare personnel improves adherence to hazardous material management guidelines, with sorting adherence rates increasing from 32.5% to 72.4% after training initiatives.
- Documentation: Maintaining , storage, and disposal is vital for compliance with regulatory requirements. Documentation practices should encompass , which are essential for reducing audit time and ensuring accountability. It is important to recognize that pharmaceutical waste is classified as universal waste and must be managed separately from biohazard waste, which varies by state, such as the 90-day storage limit in Michigan.
- Partnerships: , such as Superior Medical Disposal, ensures that hazardous materials are transported and disposed of in accordance with regulations. Engaging with reputable partners can streamline processes and enhance compliance readiness.
- Emergency Procedures: Establishing and communicating or incidents involving hazardous pharmaceutical materials is critical. Preparedness for emergencies is essential in mitigating the impact of hazardous drug exposure incidents.
By following these strategies, healthcare facilities can effectively manage hazardous pharmaceutical waste disposal, ensuring safety and compliance while minimizing environmental impact.
Conclusion
Healthcare facilities hold a pivotal responsibility in the safe disposal of hazardous pharmaceutical waste, which transcends mere regulatory compliance. By grasping the complexities of hazardous waste management, healthcare providers can safeguard public health and mitigate environmental risks linked to improper disposal practices. The significance of adhering to established guidelines is paramount, as it protects both human health and the ecosystem.
This article has underscored essential practices for managing hazardous pharmaceutical waste. Key practices include:
- Accurately identifying and classifying waste
- Complying with regulatory requirements
- Implementing effective disposal strategies
Through comprehensive staff training, diligent documentation, and partnerships with licensed waste management firms, healthcare establishments can markedly improve their waste management protocols. The dangers of improper disposal, such as environmental contamination and public health threats, further highlight the urgency of these measures.
Ultimately, the onus of managing hazardous pharmaceutical waste rests with healthcare providers, who must prioritize the adoption of best practices to safeguard both communities and the environment. As the healthcare landscape evolves, so too must the strategies for waste disposal. Embracing comprehensive waste management practices is not only a legal obligation but also a moral imperative, ensuring a safer and healthier future for all.
Frequently Asked Questions
What is hazardous pharmaceutical waste?
Hazardous pharmaceutical waste refers to any pharmaceutical item considered dangerous due to its potential to harm human health or the environment. This includes expired medications, unused drugs, and materials containing hazardous substances like chemotherapy agents and controlled substances.
How does the EPA classify hazardous pharmaceutical materials?
The Environmental Protection Agency (EPA) classifies hazardous pharmaceutical materials based on characteristics such as ignitability, corrosivity, reactivity, and toxicity. For example, substances that can easily ignite or are corrosive to human tissue fall under these classifications.
Why is understanding hazardous pharmaceutical waste classifications important for healthcare establishments?
Understanding these classifications is crucial for healthcare establishments to ensure proper management and disposal of waste. Mismanagement can lead to severe legal repercussions and environmental damage, and improper disposal practices contribute to millions of deaths worldwide due to unmanaged medical refuse.
What are the legal requirements for establishments producing medical refuse?
All establishments producing medical refuse are legally required to have their sharps and biohazard materials removed from their premises every 90 days or less. They must also obtain a Certificate of Registration, which expires every three years, with varying fees based on the type of establishment.
What are the consequences of improper disposal of hazardous pharmaceutical waste?
Improper disposal of hazardous pharmaceutical waste can lead to severe legal repercussions, environmental damage, and contributes to an estimated 5.2 million deaths annually worldwide due to unmanaged medical refuse.
How does proper management of hazardous pharmaceutical waste benefit public health and the environment?
Adhering to hazardous waste classifications and regulations protects public health and ensures compliance with legal standards, allowing healthcare establishments to operate within legal frameworks while safeguarding the environment.
