Introduction
Understanding the complexities of biomedical waste is essential for healthcare facilities committed to safeguarding public health and the environment. This article explores the definition, types, and regulatory frameworks related to biomedical waste, emphasizing the significant risks linked to improper disposal. As healthcare regulations continue to evolve, facilities must consider how to ensure compliance while effectively addressing the diverse challenges presented by biomedical refuse.
Define Biomedical Waste: Key Characteristics and Types
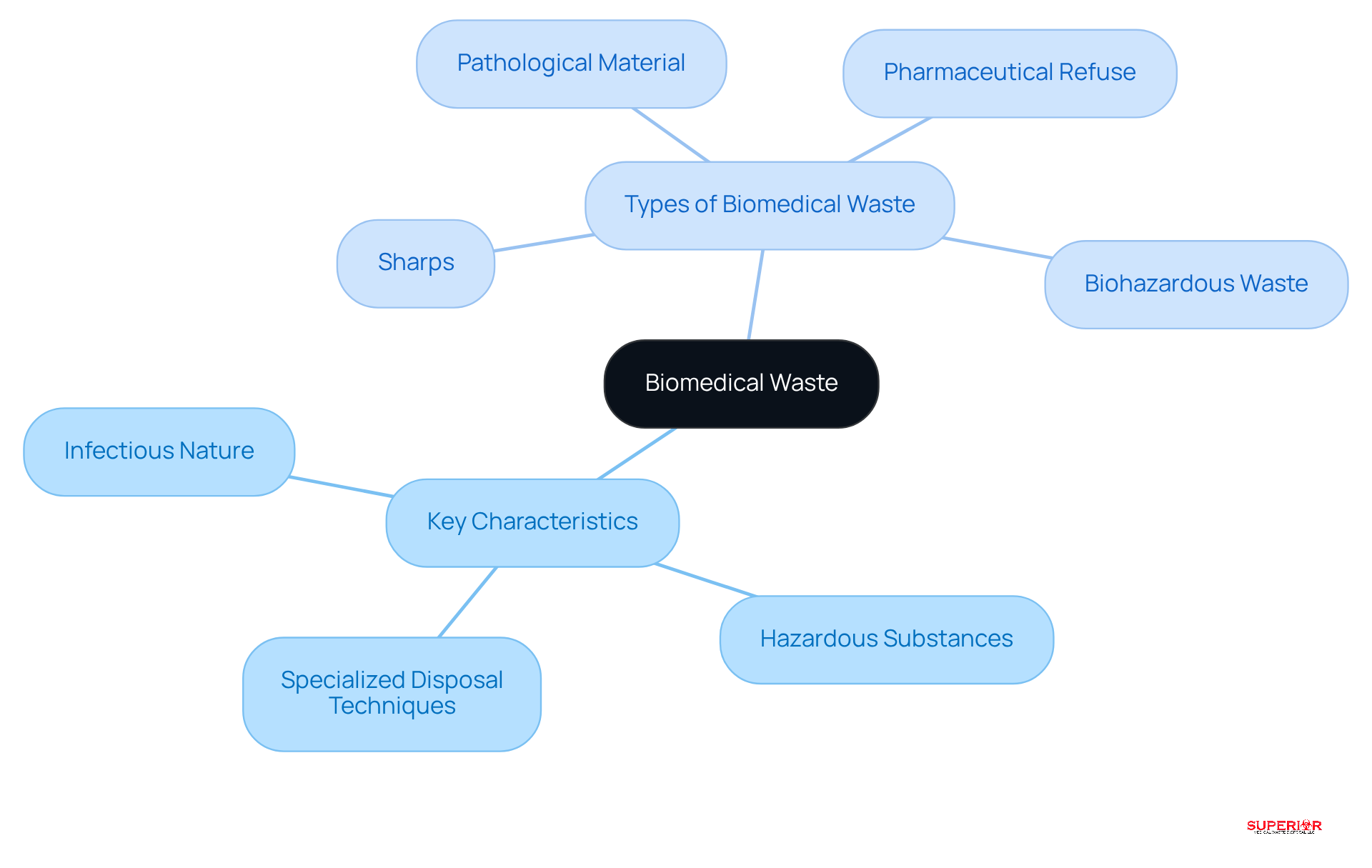
The encompasses any material generated during the diagnosis, treatment, or immunization of humans or animals, as well as during research activities, which is also commonly known as biomedical refuse. This type of refuse can pose significant hazards and is categorized based on its potential risks to public health and the environment. Key characteristics of medical refuse include its infectious nature, the presence of , and the necessity for specialized disposal techniques.
Common types of biomedical waste include:
- Sharps: Items like needles and scalpels that can cause injuries and transmit infections.
- Pathological Material: Human tissues and organs that require careful handling to prevent contamination.
- Pharmaceutical Refuse: Expired or unused medications that can be harmful if not disposed of properly. offers specialized pharmaceutical handling and removal services, ensuring compliance with and best practices.
- Biohazardous Waste: Contaminated materials that pose a risk of infection or disease transmission.
Understanding these characteristics is crucial for healthcare facilities to implement . Alarmingly, one in three healthcare establishments worldwide does not manage medical refuse safely, highlighting the urgent need for improved systems. As regulations evolve, particularly with updates expected in 2026 focusing on definitions and classifications, facilities must prioritize compliance with to mitigate risks associated with . This includes adopting that align with current standards and best practices, such as the provided by Superior Medical Refuse Management.
Contextualize Biomedical Waste: Importance and Regulatory Framework
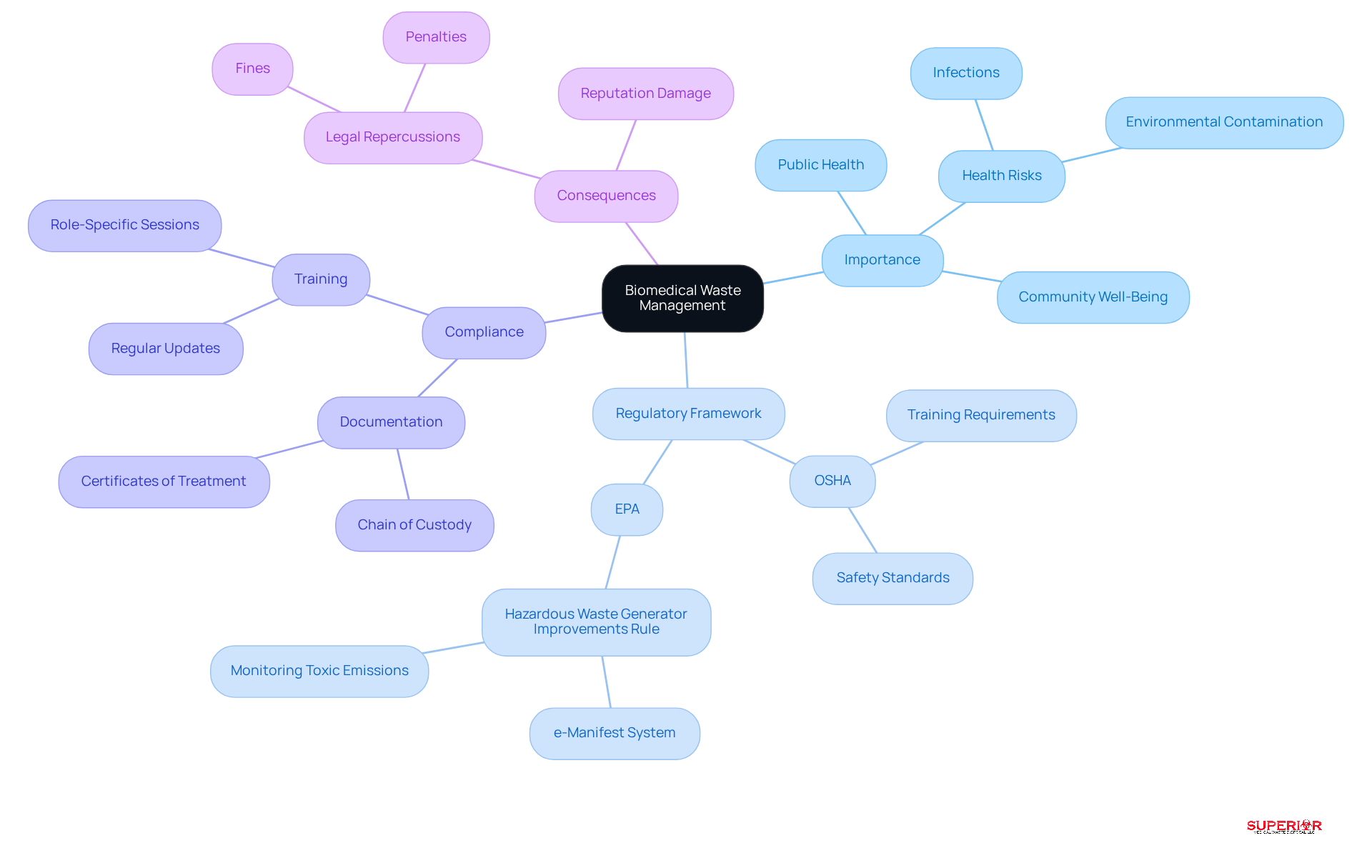
Proper handling of medical refuse is crucial due to its potential to cause infections and environmental harm. established by the and the govern the management, storage, and disposal of materials according to the . These regulations aim to protect healthcare professionals, patients, and the community from the risks associated with .
For instance, the EPA’s recent amendments to the , effective March 21, 2025, underscore the necessity for adherence to . This includes the implementation of the electronic manifest (e-Manifest) system for tracking hazardous materials. Compliance with these regulations not only reduces health risks but also bolsters the reputation of .
Facilities that fail to meet these standards face severe penalties, including substantial fines and legal repercussions, which underscores the importance of robust management protocols. By adopting , healthcare organizations can significantly reduce the risks associated with medical refuse, thereby ensuring a safer environment for both staff and the community.
Trace the Origins: Historical Development of Biomedical Waste Management
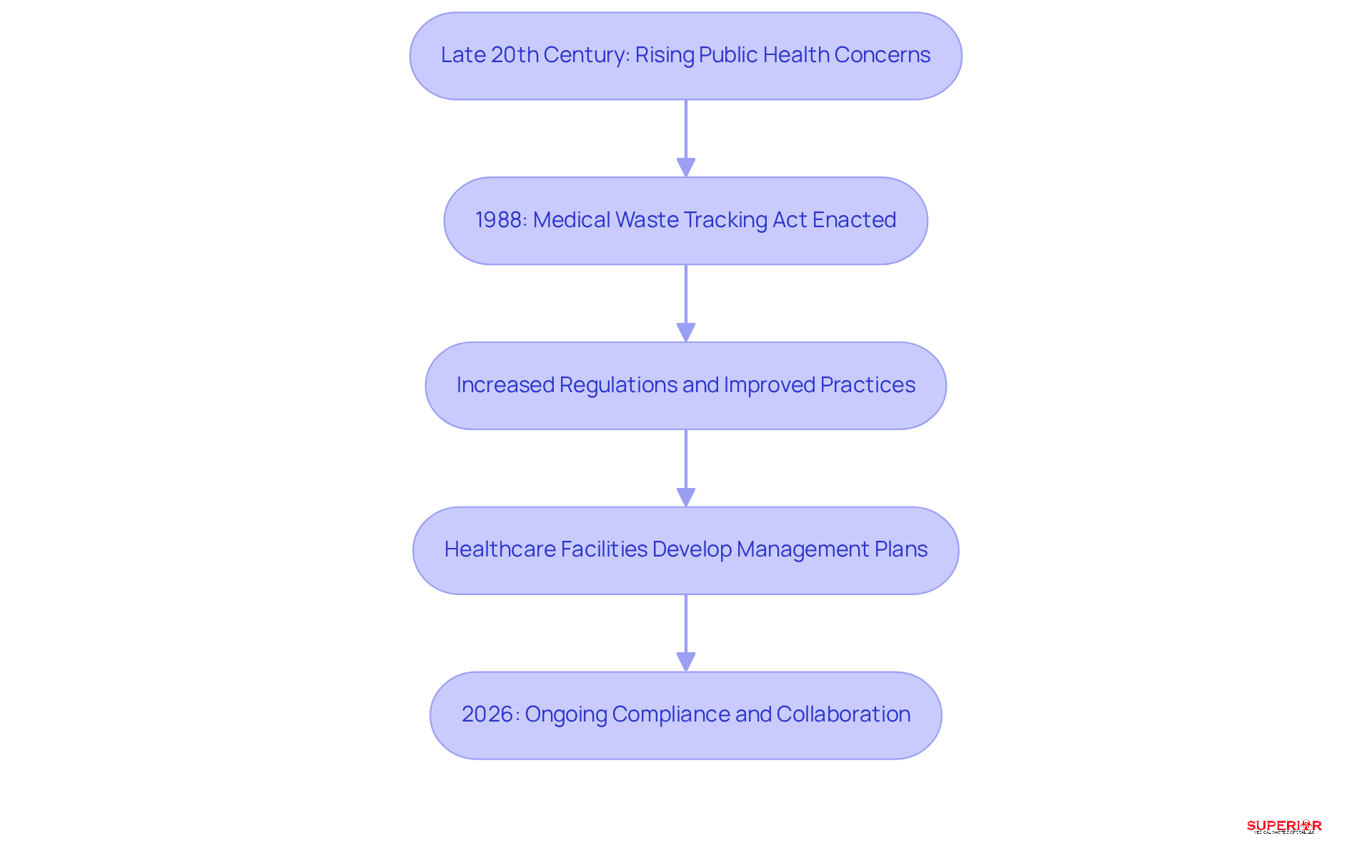
The historical progression of management gained significant momentum in the late 20th century, primarily due to rising public health concerns surrounding the handling of medical materials. A pivotal piece of legislation, the (MWTA), established federal guidelines for managing . This act was catalyzed by alarming incidents where improperly discarded materials washed ashore, leading to widespread public outcry. Its objectives included not only regulating the management of but also enhancing accountability within the healthcare sector.
Following the enactment of the MWTA, advancements in medical technology and a deeper understanding of infectious diseases have led to increasingly stringent regulations and improved disposal practices. , including notable institutions in Traverse City such as Munson, Byers Heather, and Thomas Judd, are now mandated to develop that ensure the safe handling of materials in accordance with the . These requirements reflect the lessons learned from past challenges. As of 2026, the implications of the MWTA continue to resonate, highlighting the necessity for ongoing compliance and adaptation to evolving standards in refuse management.
This evolution underscores the essential role of , refuse management providers like Superior Medical Refuse Removal, and regulatory entities in effectively addressing the complexities of . specializes in providing tailored solutions for regulated materials, including , biohazard materials, and sharps, ensuring compliance and safety for . Continuous education and adherence to regulations are vital for protecting public health and the environment.
Categorize Biomedical Waste: Types and Examples
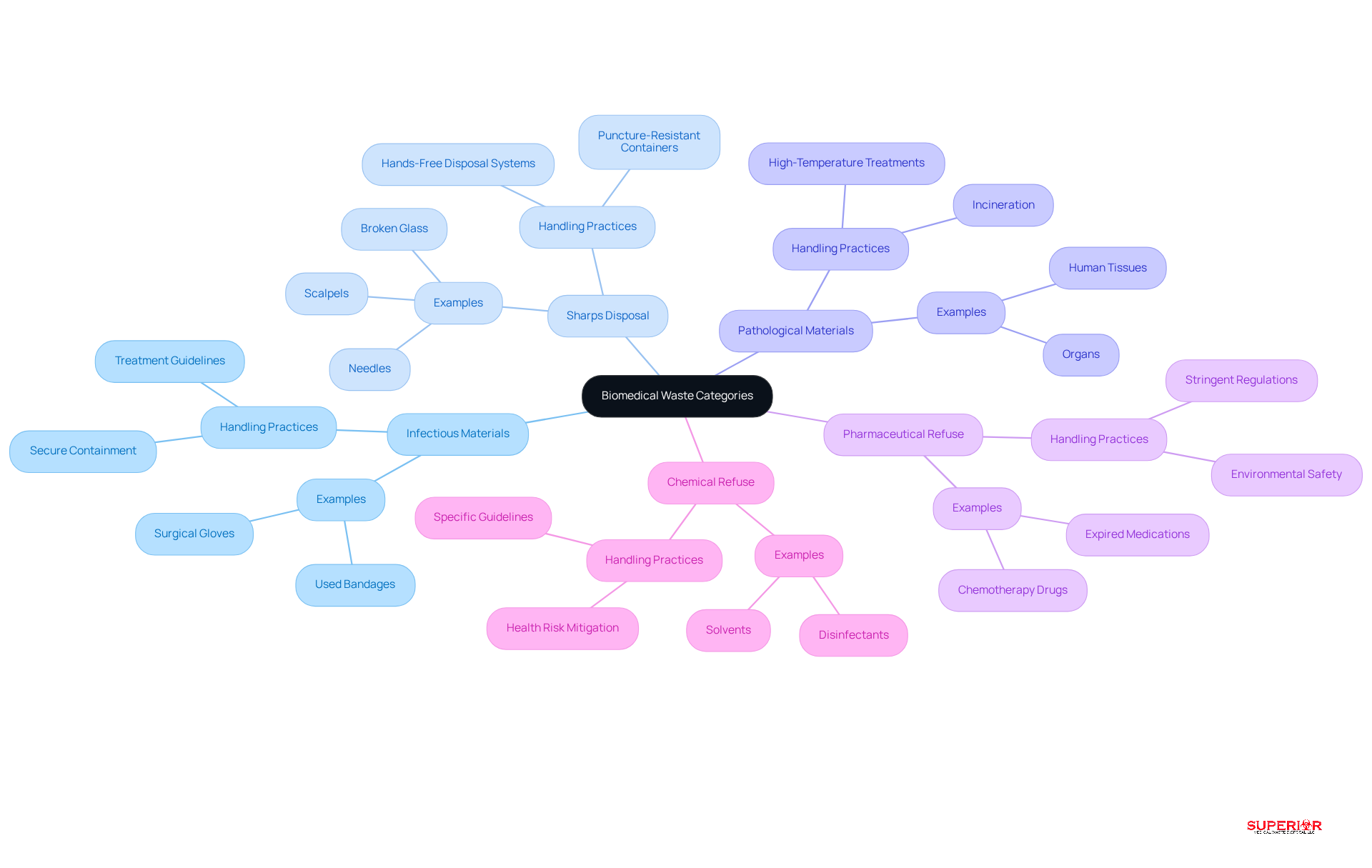
The categorizes waste into distinct types, each requiring specific handling and removal methods to mitigate risks and ensure compliance with regulatory standards. The primary categories include:
- : This category includes items contaminated with blood or other bodily fluids, such as used bandages and surgical gloves. Proper management is essential to prevent the spread of infections, with guidelines emphasizing secure containment and treatment.
- : Items capable of puncturing the skin, including needles, scalpels, and broken glass, fall under this classification. Sharps must be disposed of in puncture-resistant containers to minimize the risk of needlestick injuries and transmission of bloodborne pathogens. Current best practices advocate for hands-free disposal systems to enhance safety.
- : This category encompasses human tissues, organs, and body parts, which require meticulous handling due to their potential infectious nature. Disposal often involves incineration or high-temperature treatments to ensure complete destruction of pathogens.
- : Expired or unused medications, particularly chemotherapy drugs, are classified as . These substances must be disposed of in accordance with to prevent environmental contamination and ensure public safety.
- : Hazardous substances used in medical procedures, such as solvents and disinfectants, are classified as . Their must adhere to specific guidelines to mitigate health risks and environmental impact.
Each type of biomedical waste, as defined in the , presents unique challenges, underscoring the necessity for tailored that prioritize safety and .
Conclusion
Effective management of biomedical waste is crucial for healthcare facilities committed to protecting public health and the environment. This article underscores the urgent need for proper handling of biomedical refuse, which includes a range of materials generated during medical activities. If mishandled, this waste can pose serious risks, highlighting the necessity of adhering to established regulations and best practices.
Key points include the different types of biomedical waste – ranging from sharps to pharmaceutical refuse – and the regulatory frameworks that govern their management. The historical context of biomedical waste management illustrates how past public health crises have influenced current legislation, showcasing the ongoing evolution of disposal practices. Compliance with regulations set by organizations like OSHA and the EPA is not merely a legal requirement; it is a moral obligation to ensure the safety of healthcare professionals and the communities they serve.
Ultimately, managing biomedical waste effectively is a collective responsibility that demands continuous education, collaboration, and commitment to compliance. By prioritizing proper disposal methods and staying informed about changing regulations, healthcare facilities can significantly reduce risks and contribute to a healthier environment. Embracing these practices transcends mere compliance; it fosters a culture of safety and responsibility within the healthcare sector.
Frequently Asked Questions
What is biomedical waste?
Biomedical waste refers to any material generated during the diagnosis, treatment, or immunization of humans or animals, as well as during research activities. It is also known as biomedical refuse and can pose significant hazards to public health and the environment.
What are the key characteristics of biomedical waste?
Key characteristics of biomedical waste include its infectious nature, the presence of hazardous substances, and the necessity for specialized disposal techniques.
What are some common types of biomedical waste?
Common types of biomedical waste include:
- Sharps: Items like needles and scalpels that can cause injuries and transmit infections.
- Pathological Material: Human tissues and organs that require careful handling to prevent contamination.
- Pharmaceutical Refuse: Expired or unused medications that can be harmful if not disposed of properly.
- Biohazardous Waste: Contaminated materials that pose a risk of infection or disease transmission.
Why is it important for healthcare facilities to manage biomedical waste effectively?
Effective biomedical waste management is crucial for healthcare facilities to prevent risks to public health and the environment. Alarmingly, one in three healthcare establishments worldwide does not manage medical refuse safely, indicating an urgent need for improved systems.
What regulations are healthcare facilities expected to comply with regarding biomedical waste?
Healthcare facilities must comply with Ohio EPA regulations and adopt effective medical refuse management strategies that align with current standards and best practices, especially as regulations evolve, with updates expected in 2026 focusing on definitions and classifications.
How can facilities ensure compliance with biomedical waste management?
Facilities can ensure compliance by implementing effective medical refuse management strategies, including OSHA compliance training services provided by specialized companies like Superior Medical Refuse Management.
List of Sources
- Define Biomedical Waste: Key Characteristics and Types
- Medical waste management in the modern healthcare era: A comprehensive review of technologies, environmental impact, and sustainable practices (https://sciencedirect.com/science/article/pii/S2590123025032657)
- Global analysis of health care waste in the context of COVID-19 (https://who.int/publications/i/item/9789240039612)
- Biomedical Waste Solutions for 2026 | MedSharps (https://medsharps.com/biomedical-waste-solutions-for-2026)
- COVID-19 Biomedical Plastics Wastes—Challenges and Strategies for Curbing the Environmental Disaster (https://mdpi.com/2071-1050/14/11/6466)
- Updates on biomedical waste management during COVID-19: The Indian scenario – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9393250)
- Contextualize Biomedical Waste: Importance and Regulatory Framework
- The Critical Importance of Proper Medical Waste Management: A Deep Dive (https://glyconllc.com/the-critical-importance-of-proper-medical-waste-management-a-deep-dive)
- Regulatory Updates in Medical Waste Management | ASMAI (https://asiwaste.com/medical-waste/regulatory-updates-in-medical-waste-management)
- Biomedical Waste Solutions For 2026 | MedSharps (https://medsharps.com/blog/biomedical-waste-solutions-for-2026)
- Medical Waste Industry: Data Reports 2026 (https://wifitalents.com/medical-waste-industry-statistics)
- Trace the Origins: Historical Development of Biomedical Waste Management
- The Evolution of Medical Waste Management Regulations (https://met-bio.com/post/the-evolution-of-medical-waste-management-regulations)
- Managing 5.9 Million Tons Annually for a Sustainable Future – The Growing Challenge of Medical Waste Disposal (https://medprodisposal.com/managing-5-9-million-tons-annually-for-a-sustainable-future-the-growing-challenge-of-medical-waste-disposal)
- What Is The Medical Waste Tracking Act Of 1988? Secure Waste (https://securewaste.net/what-is-the-medical-waste-tracking-act-of-1988-everything-you-need-to-know)
- Medical Waste Tracking Act – Wikipedia (https://en.wikipedia.org/wiki/Medical_Waste_Tracking_Act)
- Categorize Biomedical Waste: Types and Examples
- Medical Waste recent news | Waste 360 (https://waste360.com/waste-collection-transfer/medical-waste)
- What Is Biomedical Waste? Key Examples (https://danielshealth.com/knowledge-center/what-is-biomedical-waste)
- The Ultimate Guide to Medical Waste Disposal in 2026 (Everything Healthcare Facilities Need to Know) (https://met-bio.com/post/medical-waste-disposal-2026-guide)
- Medical waste production at hospitals and associated factors – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7133359)
- Generation, types and impacts of biomedical waste during COVID-19: Indian context – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10834080)
