Introduction
Regulated medical waste (RMW) represents a significant concern in healthcare settings, as it includes materials that pose considerable risks of infection or injury. With medical facilities expected to generate nearly 6 million tons of clinical refuse annually by 2026, it is crucial to understand the various categories of RMW – such as sharps, pathological materials, and microbiological waste – for effective management and compliance. Navigating the complex regulatory landscape governing the safe handling and disposal of these hazardous materials presents a challenge. How can healthcare providers ensure they meet stringent regulations while also safeguarding public health and the environment?
Define Regulated Medical Waste: Key Concepts and Terminology
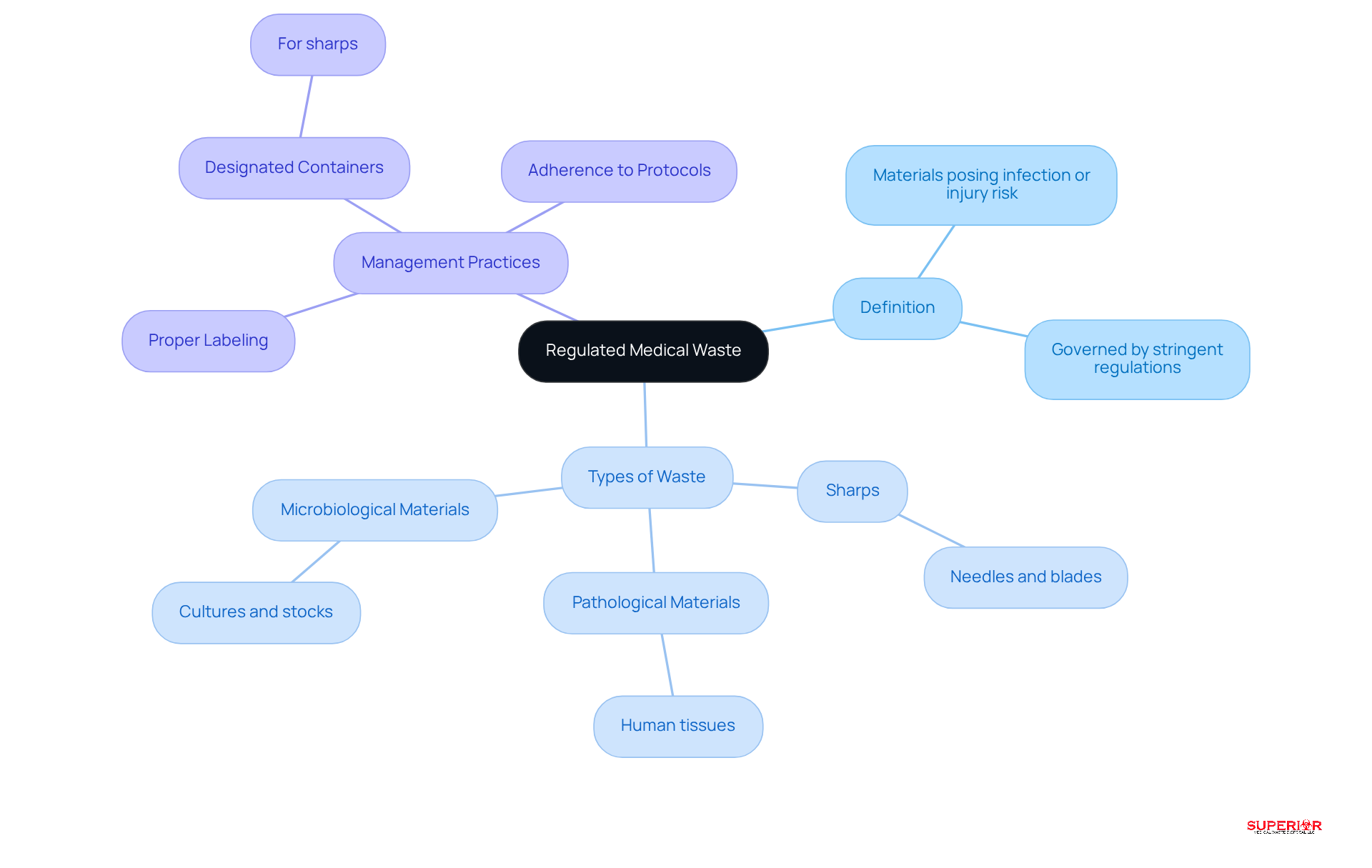
Regulated medical refuse (RMW) refers to materials generated in healthcare environments that pose a potential risk of infection or injury. Regulated medical waste includes items contaminated with blood, bodily fluids, or other potentially infectious substances, commonly known as biohazardous materials. RMW is governed by stringent regulations to ensure safe handling, treatment, and disposal, underscoring its critical role in public health and safety. Regulated medical waste includes key types such as:
- Sharps, which consist of needles and blades
- Pathological materials, made up of human tissues
- Microbiological materials, including cultures and stocks
In 2026, medical facilities are projected to produce approximately 5.9 million tons of clinical refuse annually, highlighting the importance of understanding RMW for effective disposal. Adhering to regulations not only protects medical staff and patients but also mitigates environmental hazards associated with improper waste management. Medical experts stress that grasping the complexities of controlled medical refuse is essential for implementing best practices in disposal management and ensuring compliance with legal standards.
Effective management practices for biohazardous refuse in hospitals include:
- The use of designated containers for sharps
- Proper labeling of refuse bags
- Strict adherence to management protocols
These practices are crucial for maintaining high standards of safety and compliance within healthcare settings.
Explore Categories of Regulated Medical Waste: Types and Classifications
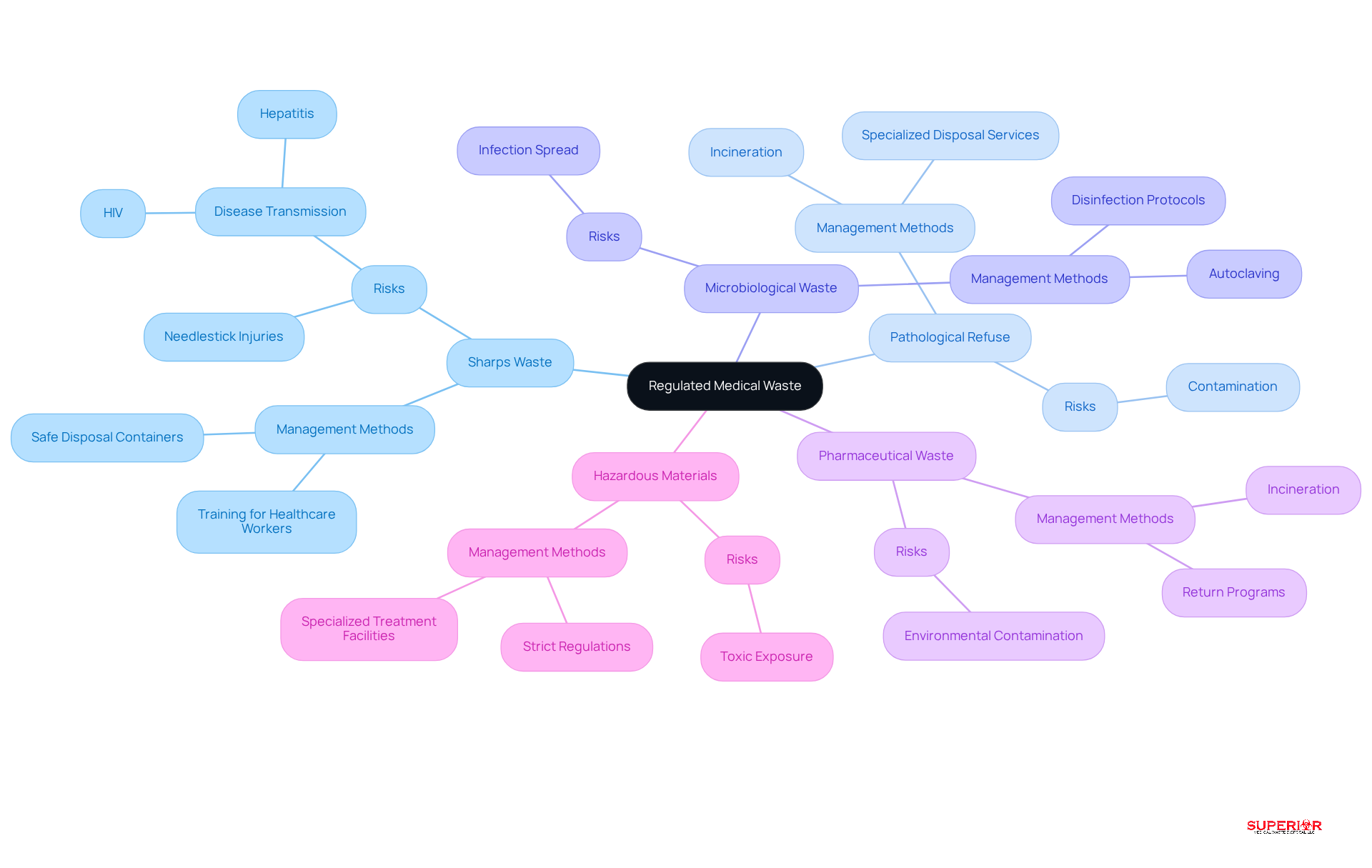
Regulated medical waste includes various categories, each necessitating specific management and disposal methods to ensure safety and compliance with regulations. The primary categories include:
-
Sharps Waste: This category includes items such as needles, blades, and broken glass, which pose significant injury risks. Improper handling can lead to needlestick injuries, potentially transmitting diseases like HIV and hepatitis.
-
Pathological Refuse: Comprising human tissues, organs, and body parts removed during surgeries or autopsies, this refuse requires careful handling to prevent contamination and ensure safe disposal.
-
Microbiological Waste: This includes cultures and stocks of infectious agents, as well as laboratory waste. Proper management is critical to prevent the spread of infections and protect public health.
-
Pharmaceutical Waste: Expired or unused medications fall into this category and must be managed using methods that prevent environmental contamination, such as incineration or specialized removal services.
-
Hazardous Materials: This encompasses any refuse that poses a danger to human health or the environment, such as chemical substances used in medical settings. Regulations mandate strict waste management techniques to mitigate these risks.
Each category is governed by specific regulations, which emphasize that regulated medical waste includes the necessity for medical facilities to adopt effective disposal strategies. Recent studies indicate that approximately 15% of medical refuse is classified as hazardous, underscoring the importance of appropriate disposal methods to minimize health risks and environmental impacts.
Understand the Importance of Proper Management: Compliance and Safety Implications
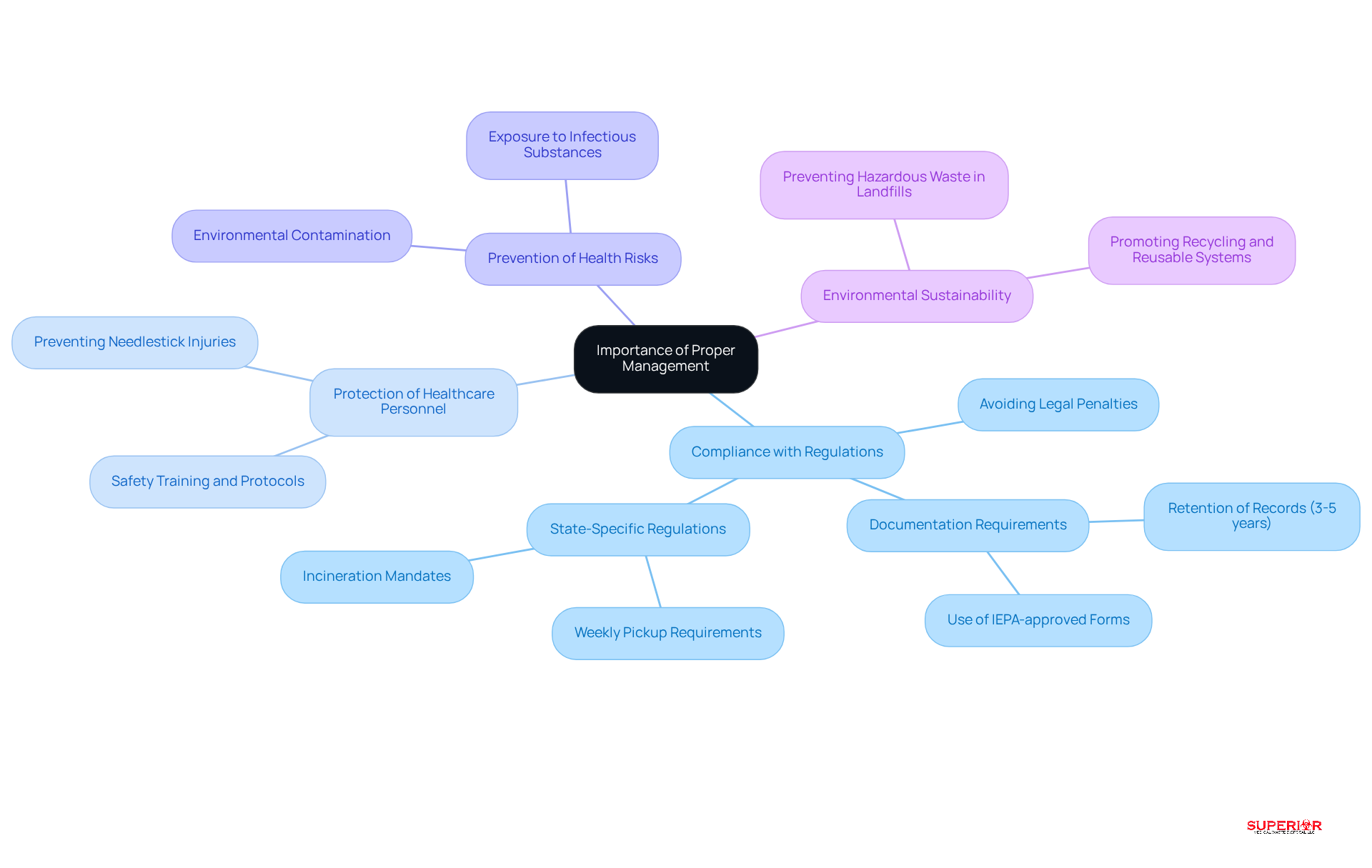
Proper handling of regulated medical waste includes crucial considerations for several reasons. It ensures compliance with federal and state regulations, as regulated medical waste includes materials that help medical facilities avoid legal penalties and fines. Additionally, efficient disposal practices protect healthcare personnel, patients, and the community from potential exposure to infectious substances. For example, improper disposal of sharps can result in needlestick injuries, which carry significant health risks. Furthermore, adhering to disposal protocols promotes environmental sustainability by preventing hazardous substances from contaminating landfills and water sources. In summary, the importance of appropriate oversight cannot be overstated, as it protects public health and upholds the integrity of healthcare operations.
Review Regulatory Framework: Federal and State Guidelines for Medical Waste Management
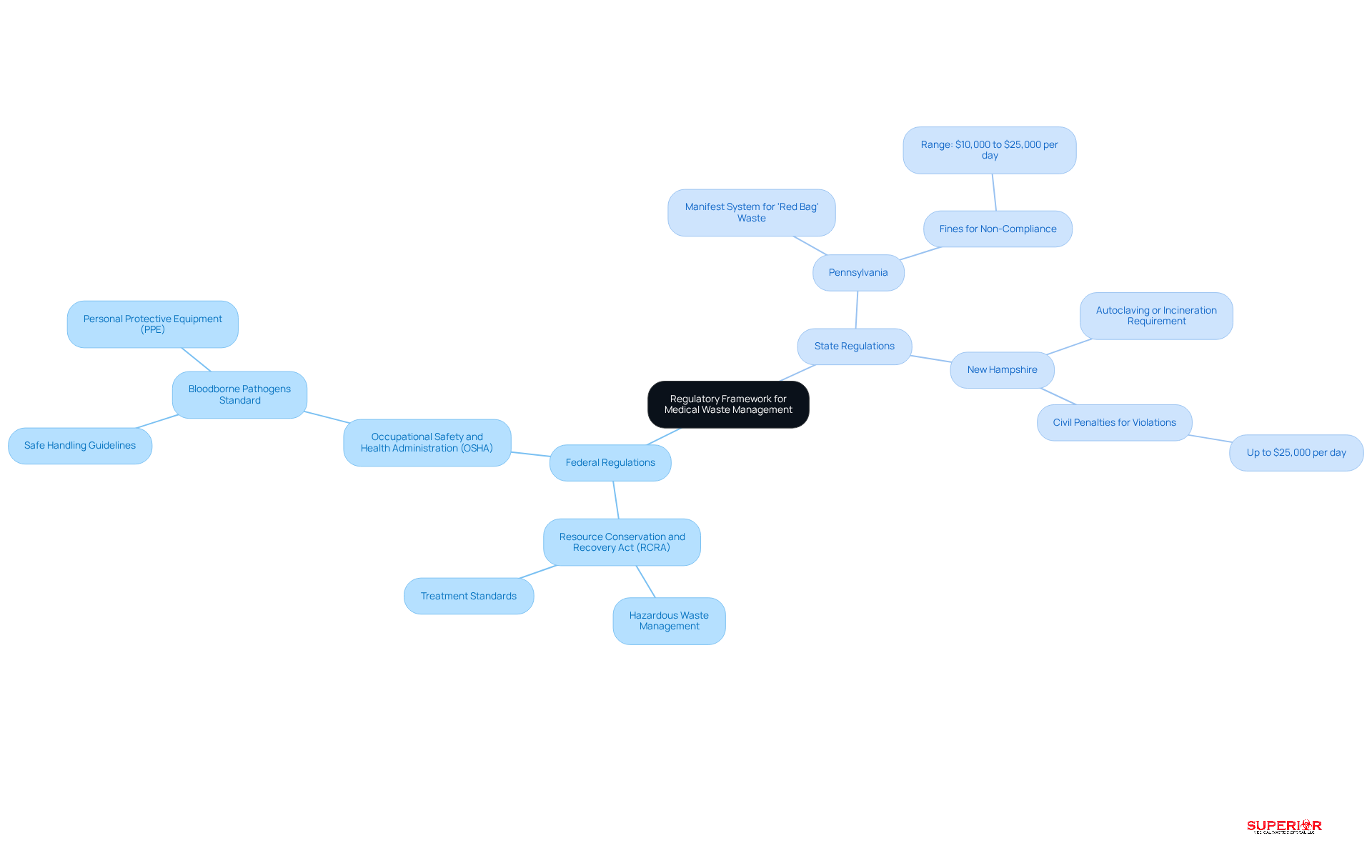
The oversight of controlled medical refuse operates within a complex framework of federal and state regulations. At the federal level, the Resource Conservation and Recovery Act (RCRA) governs the handling of hazardous materials, including medical refuse. This act ensures that facilities adhere to stringent treatment standards before disposal. Additionally, the Occupational Safety and Health Administration (OSHA) establishes guidelines for the safe handling of bloodborne pathogens, which significantly impacts the management practices for regulated medical waste.
State regulations introduce further complexity, as each state enforces specific guidelines for the management, storage, and transportation of medical materials. For example:
- Pennsylvania requires a manifest system for all ‘red bag’ materials, with fines for non-compliance ranging from $10,000 to $25,000 per day.
- New Hampshire mandates that medical refuse be autoclaved or incinerated before landfill disposal, imposing civil penalties that can reach $25,000 per day for violations.
Healthcare facilities must remain vigilant in monitoring both federal and state regulations to ensure compliance and mitigate potential legal repercussions. Recent updates, effective March 21, 2025, have clarified compliance targets for 2026, particularly emphasizing the need for medical facilities to review their pharmaceutical disposal protocols in light of stricter enforcement measures.
Understanding this regulatory environment is essential for effective refuse management and operational success in healthcare settings. Facilities that successfully navigate these complexities can minimize risks associated with improper waste disposal, safeguard public health, and ensure compliance with environmental standards.
Conclusion
Regulated medical waste (RMW) represents a range of materials produced in healthcare settings that pose significant risks to public health. Understanding the nature and categories of RMW – such as sharps, pathological materials, microbiological waste, and hazardous substances – is essential for effective management and compliance with stringent regulations. Proper handling and disposal of this waste not only protect healthcare workers and patients but also safeguard the environment from potential contamination.
This article underscores the necessity of adhering to best practices in the management of regulated medical waste. Key insights include:
- The need for designated containers
- Accurate labeling
- Strict compliance with both federal and state regulations
These practices are crucial for minimizing health risks associated with improper disposal and for avoiding legal penalties that may arise from non-compliance. As medical facilities brace for an expected increase in waste production, the urgency of implementing effective disposal strategies becomes even more critical.
Ultimately, the importance of regulated medical waste management cannot be overstated. It plays a vital role in ensuring public health safety, environmental sustainability, and operational integrity within healthcare facilities. By emphasizing the need for compliance and best practices, we contribute to a safer healthcare environment and foster a culture of responsibility in waste management. Engaging with the complexities of RMW not only protects individuals but also upholds the standards necessary for effective healthcare delivery.
Frequently Asked Questions
What is regulated medical waste (RMW)?
Regulated medical waste refers to materials generated in healthcare environments that pose a potential risk of infection or injury, including items contaminated with blood, bodily fluids, or other potentially infectious substances.
What are the key types of regulated medical waste?
The key types of regulated medical waste include sharps (needles and blades), pathological materials (human tissues), and microbiological materials (cultures and stocks).
Why is the management of regulated medical waste important?
Proper management of regulated medical waste is crucial for protecting medical staff and patients, as well as mitigating environmental hazards associated with improper waste management.
What is the projected amount of clinical refuse produced by medical facilities in 2026?
In 2026, medical facilities are projected to produce approximately 5.9 million tons of clinical refuse annually.
What are some effective management practices for biohazardous waste in hospitals?
Effective management practices include using designated containers for sharps, properly labeling refuse bags, and strictly adhering to management protocols.
How does adherence to regulations impact healthcare settings?
Adhering to regulations helps maintain high standards of safety and compliance within healthcare settings, ensuring safe handling, treatment, and disposal of regulated medical waste.
