Introduction
Managing P Listed Medications in healthcare is crucial for ensuring safety, compliance, and environmental responsibility. As healthcare facilities face the complexities of handling these hazardous substances, it is essential to understand best practices for their management. With regulations constantly evolving and the risks associated with non-compliance, healthcare providers must be prepared to tackle these challenges effectively. This article explores strategies for managing P Listed Medications, emphasizing the significance of:
- Adhering to regulations
- Providing staff training
- Implementing safe disposal methods
to safeguard public health and the environment.
Define P Listed Medications and Their Importance in Healthcare
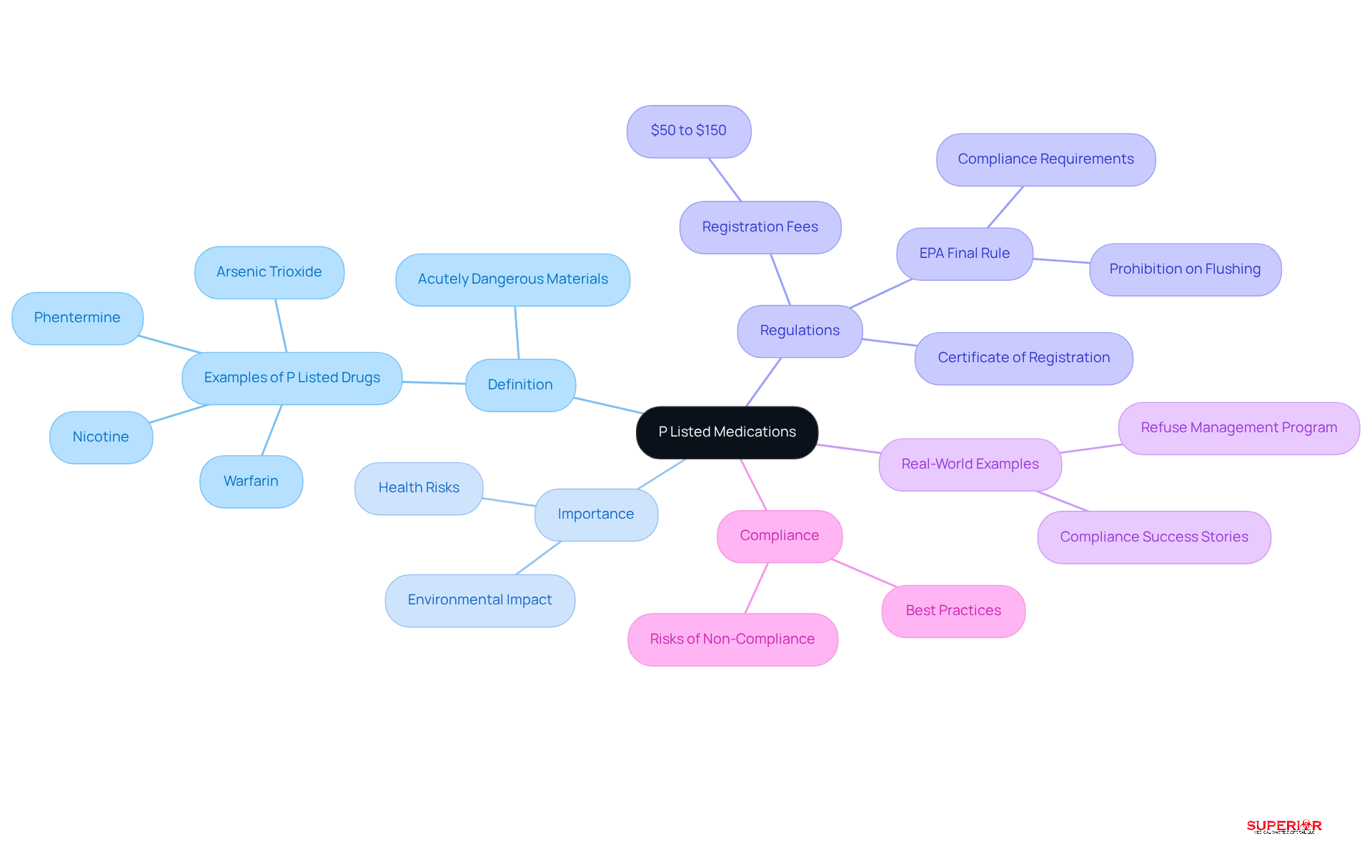
P Categorized Pharmaceuticals, as defined by the Environmental Protection Agency (EPA), include drugs considered acutely dangerous materials, such as warfarin and nicotine. These medications pose significant risks due to their potential to cause , even in minimal doses. In 2026, the handling of P Classified Drugs in healthcare is underscored by the presence of 15 such substances commonly found in pharmacy and healthcare environments, necessitating strict disposal management practices.
Correct identification and categorization of p listed medications are essential for healthcare facilities to comply with dangerous material regulations, thereby safeguarding public health and the environment. Facilities must secure a Certificate of Registration, required for any establishment generating medical byproducts, which expires every three years. Registration fees range from $50 to $150, depending on the facility type. The execution of the EPA’s Final Rule has clarified the handling of hazardous pharmaceutical materials, prohibiting the disposal of these substances via drains or toilets, a practice that previously posed considerable environmental risks.
Real-world examples illustrate the importance of effective management of hazardous materials for p listed medications. For instance, a healthcare facility that implemented a comprehensive refuse management program saw a significant reduction in compliance violations and improved operational efficiency. Facilities adhering to the Resource Conservation and Recovery Act (RCRA) regulations not only mitigate risks associated with improper disposal but also enhance their operational efficiency. Healthcare professionals stress that non-compliance with these regulations can result in enforcement actions and potential penalties, emphasizing the critical need for awareness and adherence to waste management protocols.
In summary, understanding and managing P Classified Medications is crucial for healthcare facilities in 2026, as it directly influences compliance, safety, and environmental sustainability.
Understand Regulatory Compliance for P Listed Medications
Healthcare facilities must comply with stringent regulations concerning the management of p listed medications, as outlined by the Resource Conservation and Recovery Act (RCRA). This legislation classifies p listed medications as , necessitating their removal from standard disposal streams. Key requirements include:
- Accurate labeling
- Secure storage
- Environmentally sound disposal methods
All aimed at mitigating contamination risks and protecting public health.
[Superior Medical Waste Disposal](https://superiorwastedisposal.com) enhances compliance and expertise in managing these [hazardous materials](https://www.superiorwastedisposal.com/10-reasons-healthcare-facilities-need-a-confidential-paper-shredder/) through its extensive network of strategic partnerships. Facilities are required to conduct regular audits and provide comprehensive staff training to ensure ongoing adherence to these regulations. By implementing robust resource management protocols, organizations not only meet legal standards but also contribute to broader public health initiatives by preventing drug misuse and environmental degradation.
Implement Best Practices for Safe Handling and Disposal
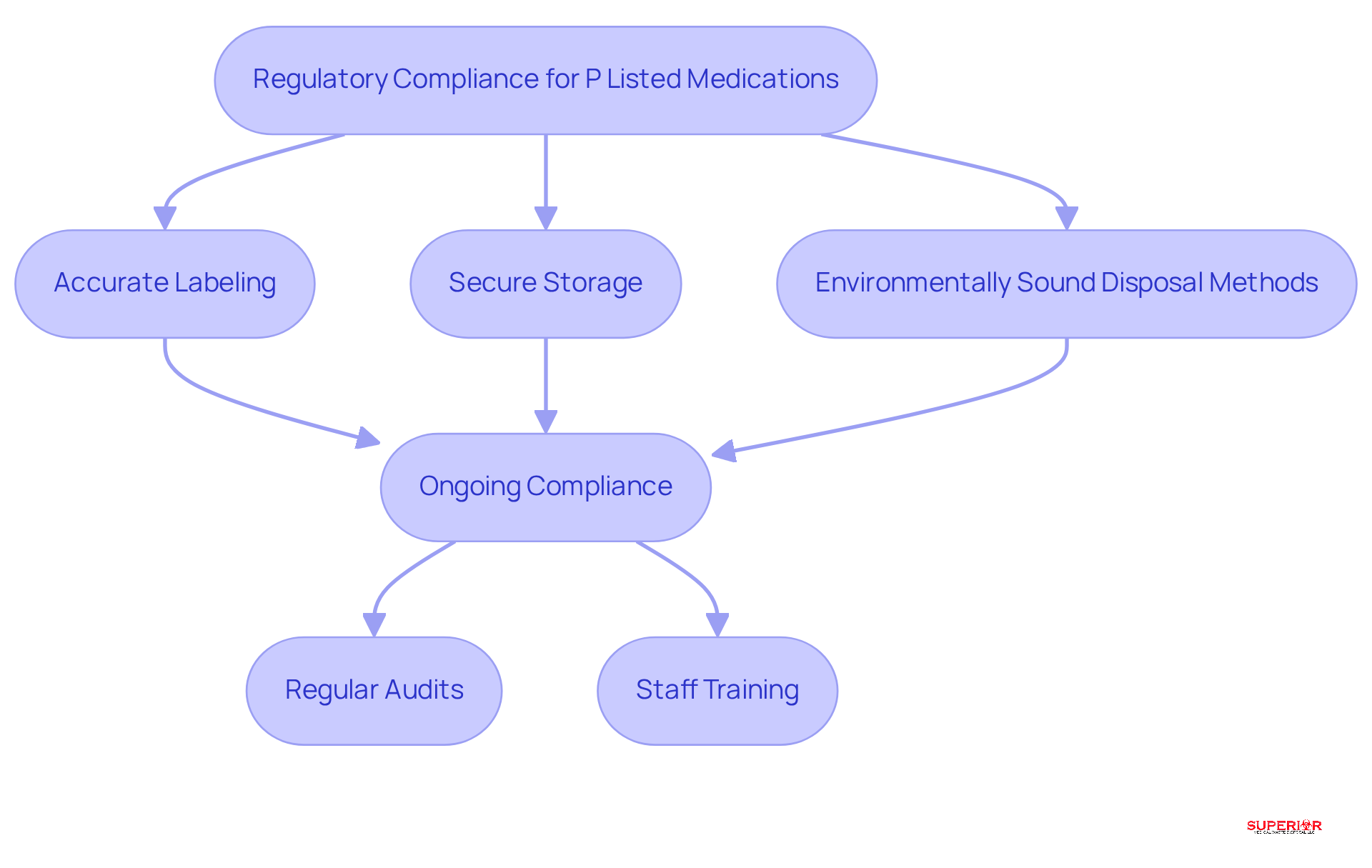
To effectively manage P Listed Medications, healthcare facilities should adopt several best practices:
- Segregation: Clearly separate P Categorized Drugs from non-hazardous materials to prevent cross-contamination. This practice not only complies with but also minimizes the risk of accidental exposure.
- Labeling: Ensure that containers holding P listed medications are clearly marked for appropriate identification and handling. Accurate labeling is crucial for compliance and facilitates efficient hazardous waste management.
- Secure Storage: Store P listed medications in a locked, designated area to prevent unauthorized access. This protects both staff and patients from potential hazards associated with these substances.
- Training: Conduct regular training sessions for staff on the proper handling and disposal procedures for P listed medications. Continuous education ensures that all personnel are informed about the latest regulations and best practices.
- Disposal Methods: Utilize certified hazardous waste disposal services, such as Superior Medical Waste Disposal, to ensure compliant disposal of P listed medications. Engaging certified services meets legal requirements and safeguards both the environment and public health.
Implementing these practices not only ensures compliance with regulations but also enhances safety for staff and patients, ultimately contributing to improved healthcare outcomes.
Prioritize Staff Training and Education on P Listed Medications
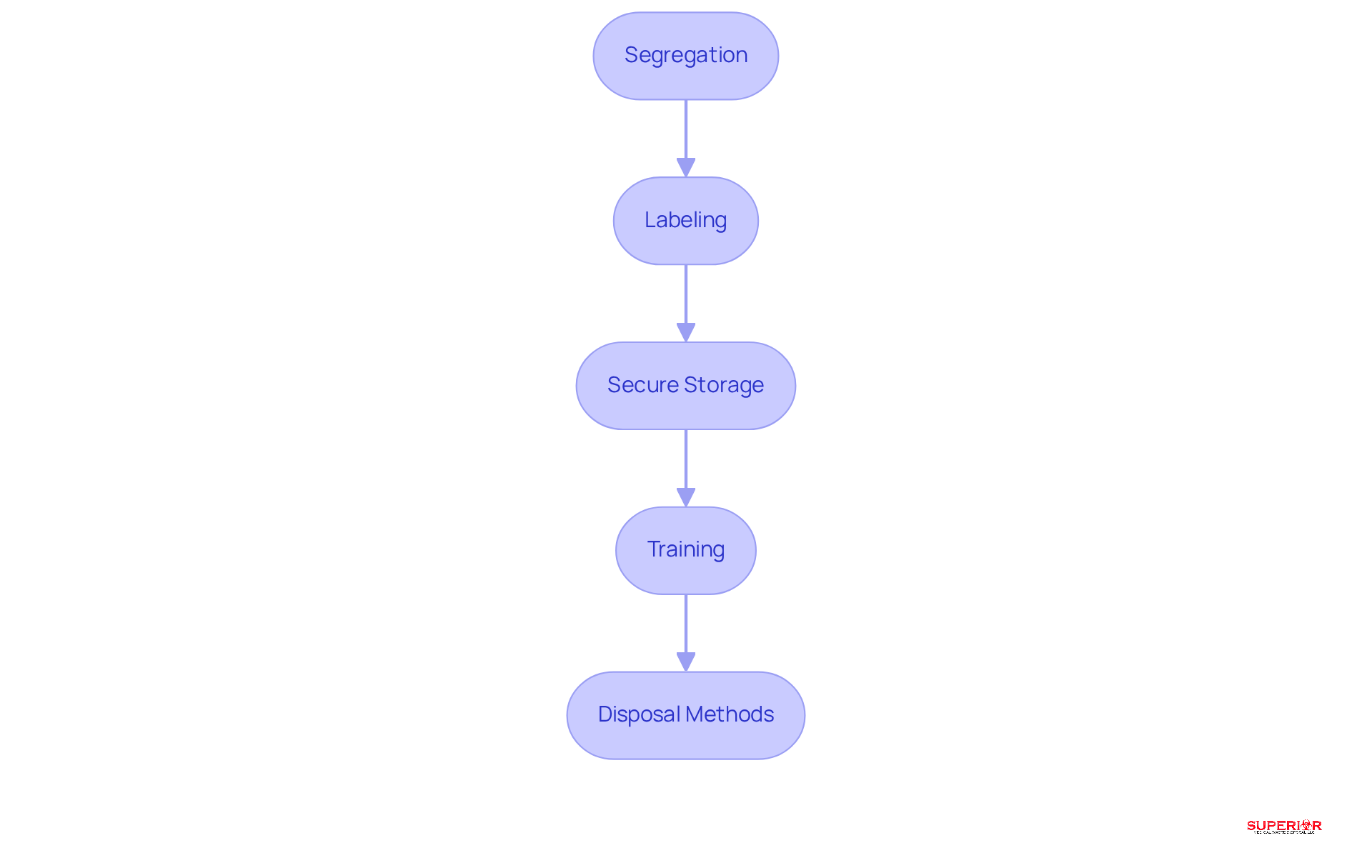
Effective training programs are crucial for the proper management of P classified substances in healthcare settings. Facilities should concentrate on several key areas to ensure that staff are adequately prepared:
- Identification: Staff must be trained to accurately recognize [P Classified Drugs](https://www.superiorwastedisposal.com/best-practices-for-managing-p-listed-waste-drugs-in-healthcare/) and understand the associated risks, which is essential for safe handling.
- Handling Procedures: Comprehensive training on safe handling techniques is vital to minimize exposure and prevent accidents. This includes the use of (PPE) and proper storage methods.
- Emergency Protocols: Establishing clear procedures for responding to spills or accidents involving p listed medications is critical. Staff should be well-versed in emergency response protocols to effectively mitigate risks.
- Regulatory Updates: Keeping staff informed about the latest regulations and best practices in dangerous material management is essential for compliance. Regular training sessions, workshops, and refresher courses can reinforce knowledge and ensure adherence to evolving standards.
Research indicates that well-trained personnel are significantly less likely to make errors that could lead to dangerous situations. For example, a comprehensive training program can enhance competencies by over 30%, resulting in improved safety and operational efficiency. By prioritizing education on hazardous waste management, healthcare facilities can cultivate a culture of safety and compliance, ultimately protecting both staff and patients from the dangers associated with improper handling of hazardous materials.
Conclusion
Understanding the management of P listed medications is crucial for healthcare facilities, as it directly influences compliance, safety, and environmental stewardship. Effectively handling these hazardous substances not only ensures adherence to regulatory frameworks but also protects public health and minimizes environmental risks associated with improper disposal.
This article highlights key practices for managing P classified medications, including:
- Accurate labeling
- Secure storage
- Robust staff training
The significance of regulatory compliance, as outlined by the Resource Conservation and Recovery Act, cannot be overstated. By implementing best practices and engaging certified disposal services, healthcare facilities can significantly reduce risks, enhance operational efficiency, and foster a culture of safety.
In conclusion, prioritizing the management of P listed medications is essential for safeguarding both healthcare staff and patients. By committing to comprehensive training and strict adherence to regulations, healthcare facilities can contribute to a safer environment and uphold their responsibility in public health. The call to action is clear: investing in proper management strategies is not merely a regulatory obligation but a critical step toward ensuring the well-being of the community and the environment.
Frequently Asked Questions
What are P Listed Medications?
P Listed Medications are pharmaceuticals categorized by the Environmental Protection Agency (EPA) as acutely dangerous materials, which can cause severe health issues even in minimal doses. Examples include drugs like warfarin and nicotine.
Why are P Listed Medications important in healthcare?
They pose significant risks to public health and the environment, necessitating strict disposal management practices to prevent health hazards and environmental contamination.
How many P Classified Drugs are commonly found in healthcare settings?
There are 15 P Classified Drugs commonly found in pharmacy and healthcare environments.
What is required for healthcare facilities handling P Listed Medications?
Healthcare facilities must secure a Certificate of Registration to comply with dangerous material regulations, which expires every three years. Registration fees range from $50 to $150, depending on the facility type.
What does the EPA’s Final Rule state regarding the disposal of P Listed Medications?
The EPA’s Final Rule prohibits the disposal of P Listed Medications via drains or toilets, addressing significant environmental risks associated with such practices.
What are the benefits of effective management of hazardous materials for P Listed Medications?
Effective management can lead to a reduction in compliance violations, improved operational efficiency, and enhanced safety within healthcare facilities.
What are the consequences of non-compliance with regulations regarding P Listed Medications?
Non-compliance can result in enforcement actions and potential penalties, highlighting the importance of adhering to waste management protocols.
How does understanding P Classified Medications impact healthcare facilities?
Understanding and managing P Classified Medications is crucial for compliance, safety, and environmental sustainability in healthcare facilities as of 2026.
