Introduction
In healthcare, managing biohazardous waste is crucial, yet it often goes unnoticed, with significant implications for safety and compliance. Healthcare facilities must understand the various categories of biohazardous waste – ranging from sharps to pharmaceuticals – to protect both staff and patients effectively. Despite the critical nature of proper disposal methods, many medical professionals remain uncertain about their waste management practices. To address this knowledge gap, facilities need to ensure that every aspect of biohazard waste handling adheres to stringent regulatory standards.
Identify Biohazardous Waste Types for Proper Disposal
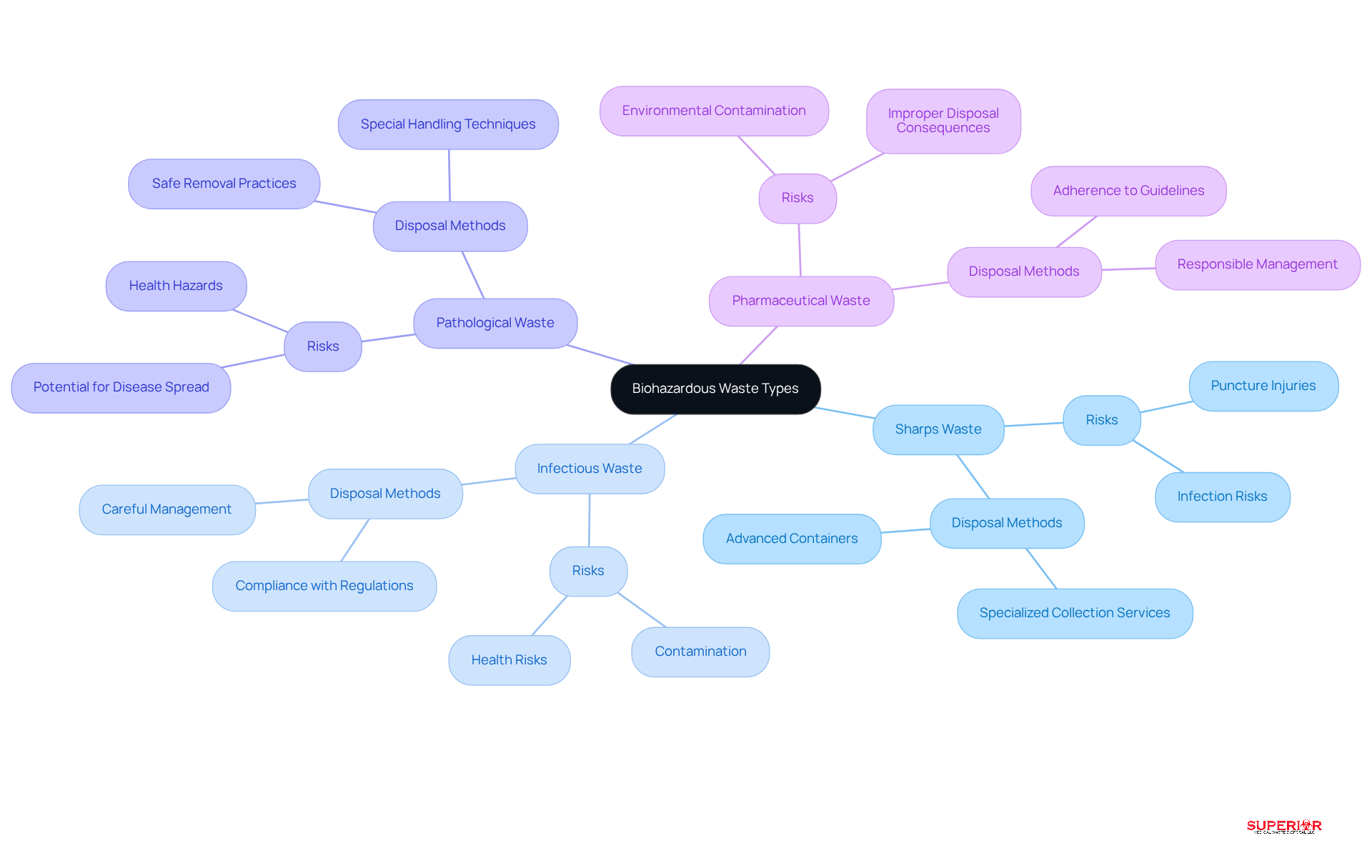
Recognizing biohazardous waste bags categories is crucial in healthcare settings to ensure safe disposal and compliance with regulations. The primary categories include:
- Sharps Waste: This category encompasses needles, scalpels, and broken glass, which present significant risks of puncture injuries and infections. Superior Medical Waste Disposal provides specialized sharps collection and disposal services, utilizing advanced containers for secure handling and disposal.
- Infectious Waste: Materials contaminated with blood or bodily fluids, such as dressings and surgical waste, fall under this category and require careful management. Our experienced team ensures compliance with all state and federal regulations in handling these materials.
- Pathological Waste: Human tissues, organs, and body parts necessitate special handling techniques due to their potential health risks. We prioritize the safe and compliant removal of these materials from your facility.
- Pharmaceutical Waste: Expired or unused medications must be disposed of according to strict regulations to prevent environmental contamination. Superior Medical Disposal is equipped to manage pharmaceutical refuse responsibly, adhering to all applicable guidelines.
Accurate identification of these waste types is essential for selecting appropriate disposal methods and biohazardous waste bags, which helps minimize exposure risks and ensures compliance with state and federal regulations. Notably, studies show that only 58% of medical professionals feel confident in safely disposing of medical and pharmaceutical waste generated in home settings, underscoring the need for enhanced training and awareness. Furthermore, 92% of medical professionals indicated a need for more training to maintain safe working conditions. Implementing effective identification practices, along with an updated Exposure Control Plan, can significantly improve safety and compliance within healthcare environments. Facilities must also recognize that serious OSHA violations can result in fines up to $16,550 per infraction, emphasizing the importance of proper disposal management practices. We recommend that all from your facility every 90 days or less, and each facility must possess a shipping document from the moment the transporter departs and a second document once the refuse is processed by the treatment center.
Choose Appropriate Biohazard Bags for Compliance and Efficiency
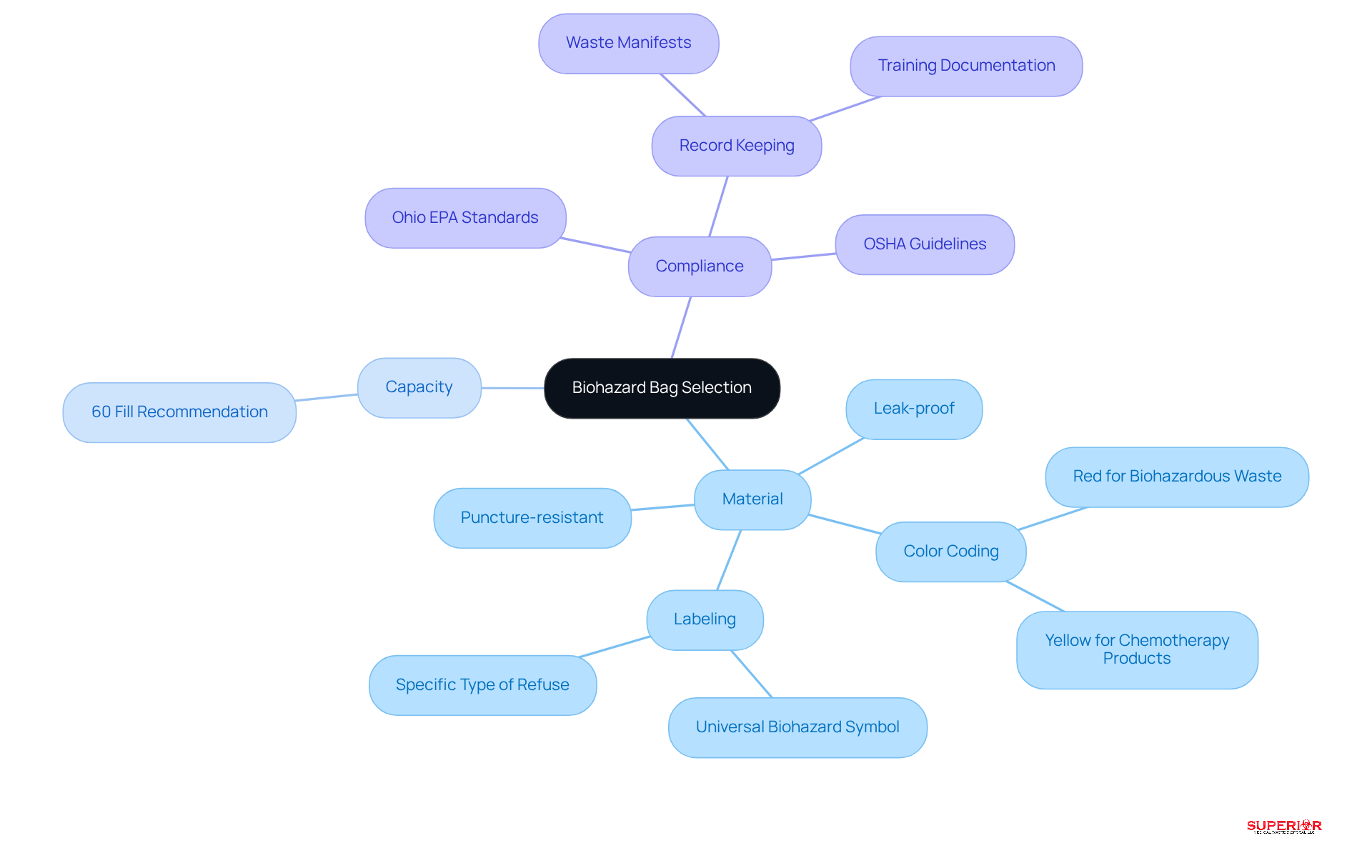
Selecting the appropriate biohazardous waste bags is essential for effective disposal management in healthcare environments. The key considerations include:
- Material: Choose bags made from puncture-resistant and leak-proof materials to prevent spills and contamination during handling and transport. Innovations in bag design, such as multi-layer constructions and antimicrobial properties, further enhance safety and hygiene. Color coding should be followed, with red bags typically designated for biohazardous waste bags, while yellow bags may be used for specific clinical items, such as chemotherapy products. This system ensures proper segregation and , including those set by the Ohio EPA. Labeling biohazardous waste bags is crucial; all bags must be clearly marked with the universal biohazard symbol and the specific type of refuse to alert staff and handlers to potential hazards. Proper labeling is crucial for maintaining safety and compliance during audits, as emphasized by OSHA.
- Capacity: Avoid overfilling bags, as this can compromise their integrity and increase the risk of exposure. It is advisable to fill bags to approximately 60% capacity, allowing for adequate headspace to prevent leaks.
Moreover, it is vital to maintain thorough record-keeping methods to ensure compliance with Ohio’s regulations regarding medical refuse generation and treatment. By following these guidelines, medical facilities can significantly improve their refuse management procedures, ensuring alignment with OSHA and CDC standards while safeguarding public health and employee safety.
Implement Safe Handling and Disposal Procedures for Biohazard Waste
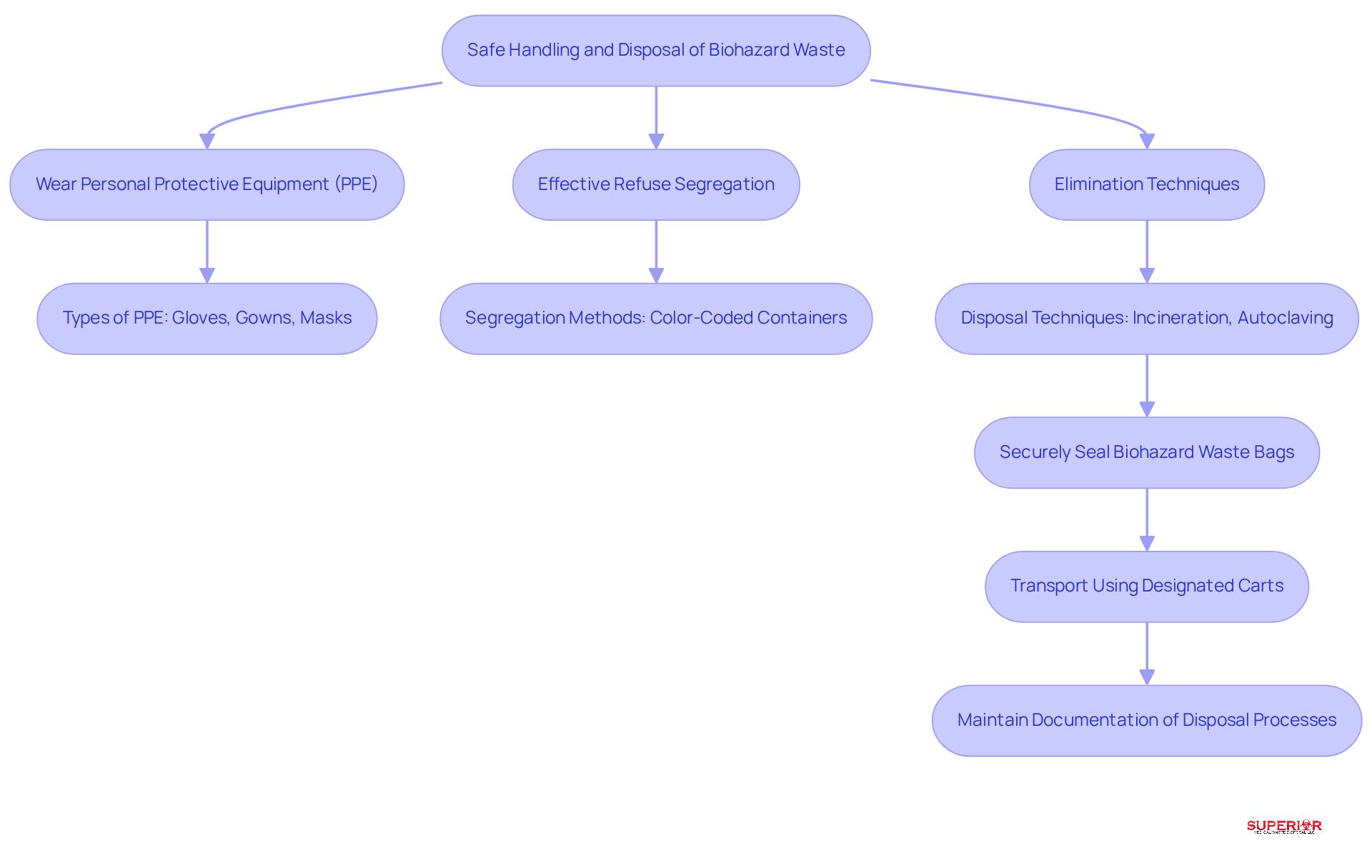
To ensure the safe handling and disposal of biohazardous waste, healthcare facilities must adopt several best practices:
- Personal Protective Equipment (PPE): Staff must wear appropriate PPE, including gloves, gowns, and masks, when handling biohazardous materials. This protective equipment minimizes the risk of exposure to infectious agents and ensures compliance with OSHA standards, which mandate initial training and annual refreshers for all employees who may come into contact with such materials.
- Refuse Segregation: Effective refuse segregation is crucial for preventing cross-contamination. Facilities should clearly designate areas for different types of waste and utilize color-coded containers for easy identification. Research indicates that facilities investing in segregation training can expect reduced waste management costs, lower contamination rates, and improved compliance scores.
Biohazardous waste bags must be securely sealed before they are transported to waste locations. Designated carts should be used to minimize exposure during transport, ensuring that refuse is handled safely and efficiently.
- Elimination Techniques: Adhering to state and federal regulations for the disposal of biohazardous materials is essential. This may involve incineration or autoclaving, depending on the material type. Facilities must maintain accurate documentation of disposal processes to comply with regulatory requirements and avoid penalties.
By implementing these protocols, medical facilities can significantly mitigate the risks associated with biohazardous materials and ensure compliance with regulatory standards, ultimately enhancing safety for both personnel and patients.
Conduct Regular Training and Compliance Audits for Staff
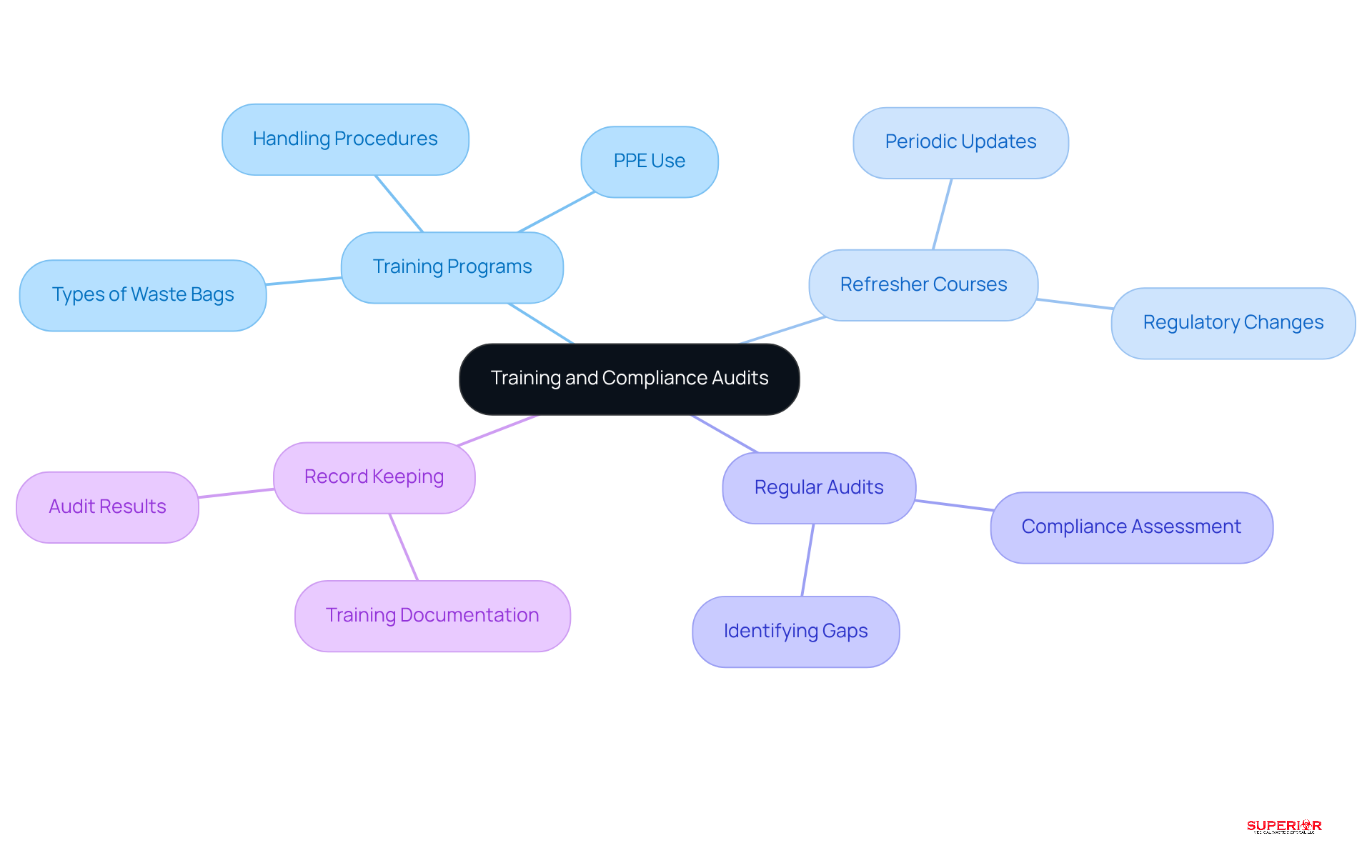
Routine training and adherence evaluations are essential for equipping healthcare personnel with the necessary knowledge for effective biohazardous material management. Prioritizing these elements fosters a culture of safety and compliance within healthcare facilities.
Training Programs: Establish that cover the various types of biohazardous waste bags, appropriate handling procedures, and the correct use of personal protective equipment (PPE). Mandatory training for all staff members ensures a uniform understanding of protocols, which is critical for maintaining safety standards.
- Implement periodic refresher courses to keep staff informed about evolving regulations and best practices. These sessions are vital for reinforcing knowledge and adapting to changes in regulatory requirements, ensuring that all personnel remain competent in their roles.
- Conduct regular audits to assess adherence to waste management protocols. These audits not only identify gaps in compliance but also provide valuable insights for enhancing training programs and operational practices, ultimately leading to improved safety outcomes.
- Maintain thorough records of training sessions and audit results. This documentation is crucial for demonstrating compliance and supporting continuous improvement efforts within the facility. It serves as a foundation for accountability and transparency in the management of biohazardous waste bags.
Conclusion
Effective management of biohazardous waste in healthcare settings is essential for ensuring safety and compliance with regulations. By identifying the various categories of biohazardous waste – such as sharps, infectious, pathological, and pharmaceutical waste – facilities can implement appropriate disposal methods and select the correct biohazard bags. This careful identification and categorization minimize exposure risks and uphold the standards set by regulatory bodies, ultimately protecting both healthcare workers and patients.
Key practices include:
- Selecting puncture-resistant and leak-proof bags
- Adhering to proper filling capacities
- Providing thorough training for staff on safe handling procedures
Regular audits and training refreshers are crucial for maintaining compliance and ensuring personnel are equipped with the latest knowledge and skills to manage biohazardous materials effectively. Emphasizing proper labeling and segregation of waste further reinforces the commitment to safety and regulatory adherence.
In conclusion, adopting best practices for biohazardous waste management is not merely a regulatory obligation; it is a critical component of a healthcare safety culture. Facilities must prioritize ongoing education and the implementation of robust waste management protocols. By doing so, healthcare institutions can significantly enhance safety outcomes, reduce the risk of contamination, and foster a responsible approach to handling biohazardous materials.
Frequently Asked Questions
What are the main categories of biohazardous waste?
The main categories of biohazardous waste are Sharps Waste, Infectious Waste, Pathological Waste, and Pharmaceutical Waste.
What is included in Sharps Waste?
Sharps Waste includes needles, scalpels, and broken glass, which pose significant risks of puncture injuries and infections.
How is Infectious Waste defined?
Infectious Waste consists of materials contaminated with blood or bodily fluids, such as dressings and surgical waste, which require careful management.
What does Pathological Waste refer to?
Pathological Waste refers to human tissues, organs, and body parts that require special handling due to potential health risks.
What is Pharmaceutical Waste and how should it be disposed of?
Pharmaceutical Waste includes expired or unused medications that must be disposed of according to strict regulations to prevent environmental contamination.
Why is accurate identification of biohazardous waste types important?
Accurate identification is essential for selecting appropriate disposal methods and biohazardous waste bags, minimizing exposure risks, and ensuring compliance with regulations.
What percentage of medical professionals feel confident in disposing of medical and pharmaceutical waste at home?
Only 58% of medical professionals feel confident in safely disposing of medical and pharmaceutical waste generated in home settings.
What do studies indicate about the need for training among medical professionals?
92% of medical professionals indicated a need for more training to maintain safe working conditions regarding waste disposal.
What are the consequences of serious OSHA violations related to biohazardous waste disposal?
Serious OSHA violations can result in fines up to $16,550 per infraction.
How often should medical refuse be removed from healthcare facilities?
Medical refuse should be removed from facilities every 90 days or less.
What documentation is required during the disposal process of biohazardous waste?
Facilities must possess a shipping document from the moment the transporter departs and a second document once the refuse is processed by the treatment center.
