Introduction
Understanding the complexities of pharmaceutical waste management is essential for healthcare facilities committed to safety and compliance. The Resource Conservation and Recovery Act (RCRA) specifies regulations that classify certain pharmaceuticals as hazardous. This makes it crucial for organizations to effectively navigate these guidelines.
With the potential for significant penalties for non-compliance, healthcare providers must ensure they are not only adhering to the rules but also implementing best practices that promote a culture of safety and environmental responsibility.
By exploring the latest strategies in RCRA pharmaceutical waste management, organizations can illuminate the path toward effective compliance and sustainable healthcare operations.
Understand RCRA Regulations for Pharmaceutical Waste
The management of hazardous materials, including pharmaceuticals, within healthcare settings is fundamentally guided by the RCRA pharmaceutical waste regulations. Understanding the specific that classify certain pharmaceuticals as hazardous is crucial for healthcare establishments.
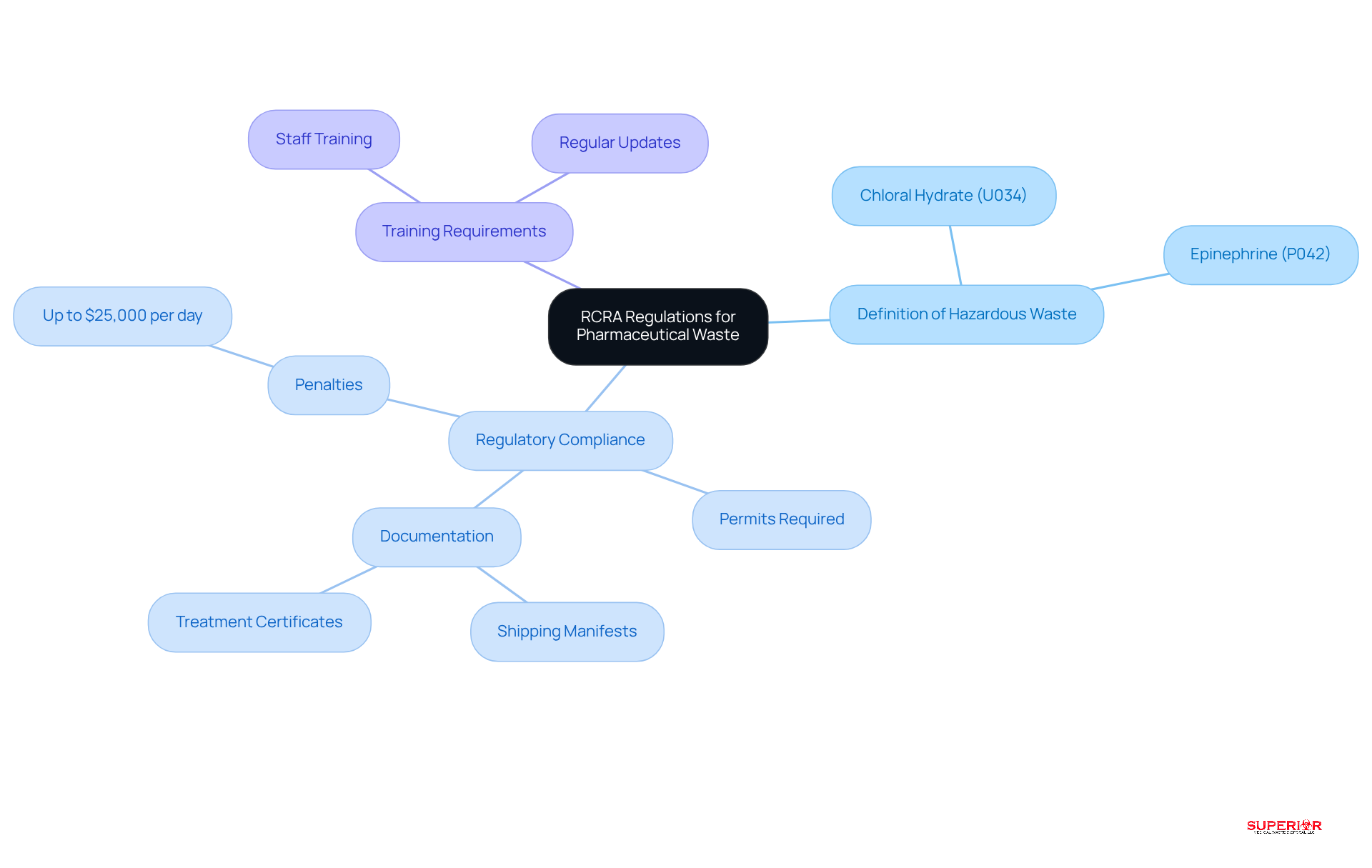
Definition of Hazardous Waste: According to RCRA, hazardous waste is defined as any waste that poses a significant threat to human health or the environment. This includes pharmaceuticals that are ignitable, corrosive, reactive, or toxic, such as chloral hydrate (classified as U034) and epinephrine (P042).
Regulatory Compliance: Healthcare establishments are required to comply with both federal and state regulations, which may necessitate obtaining permits for the management of hazardous materials. Compliance entails maintaining accurate documentation, including shipping manifests and treatment certificates, which must be retained for three to five years. Failure to comply can result in penalties of up to $25,000 per day for violations.
Training Requirements: It is essential for staff to receive [training on RCRA pharmaceutical waste regulations](https://medprodisposal.com/medical-waste-disposal-regulations-2026-state-by-state-checklist) to ensure compliance and the safe handling of hazardous pharmaceutical materials. Regular updates to training programs are necessary to keep pace with evolving regulations, particularly as medical institutions face increased scrutiny and enforcement regarding disposal definitions.
By thoroughly understanding these regulations, healthcare organizations can implement effective disposal management strategies that prioritize safety, compliance, and environmental responsibility.
Identify Types of Hazardous Pharmaceutical Waste
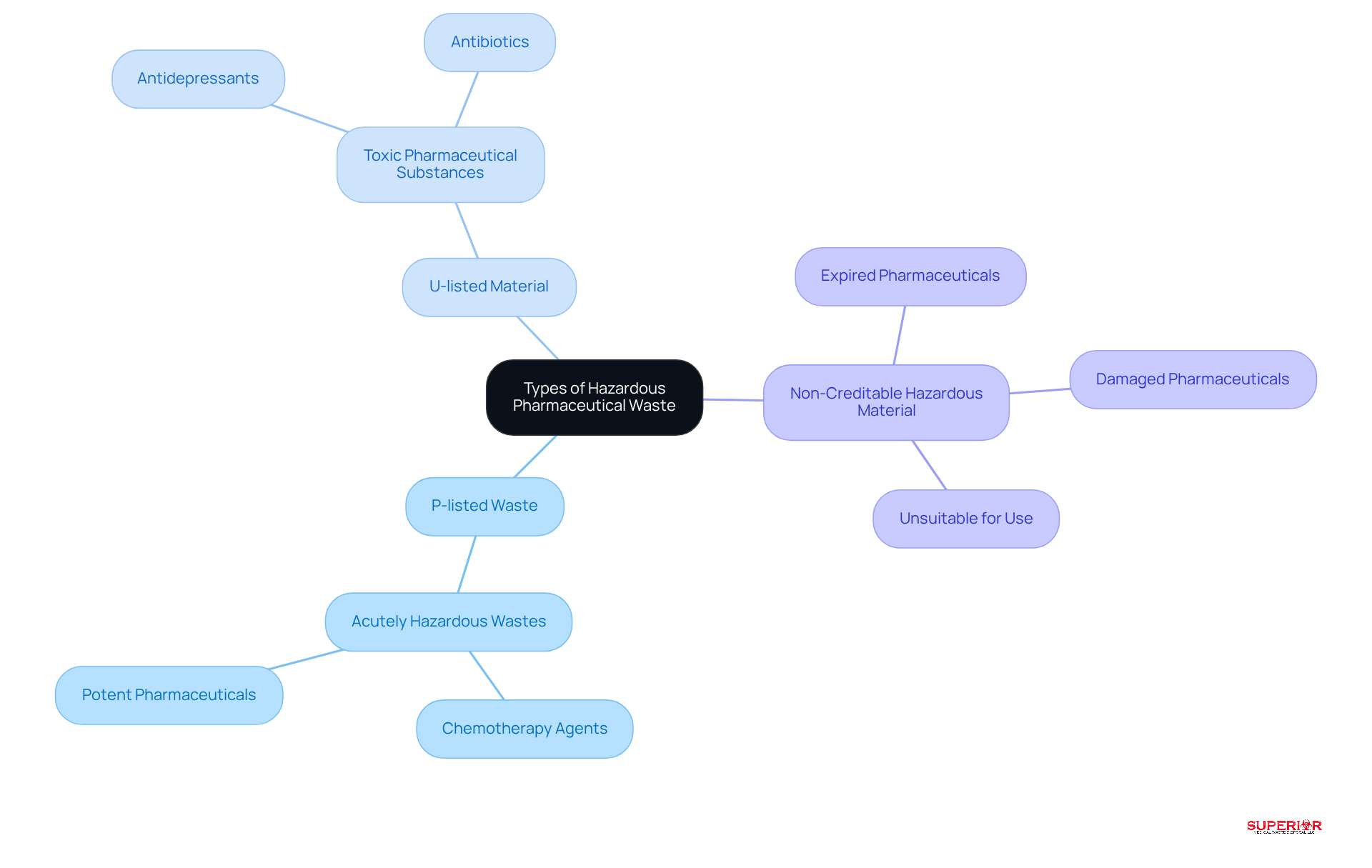
Healthcare facilities must accurately identify the types of [hazardous pharmaceutical byproducts](https://epa.gov/hwgenerators/management-hazardous-waste-pharmaceuticals) they generate to ensure compliance with RCRA pharmaceutical waste regulations. This identification is crucial for minimizing contamination risks and ensuring proper disposal. The primary categories of hazardous pharmaceutical byproducts include:
- P-listed Waste: This category encompasses acutely hazardous wastes, including specific chemotherapy agents and other potent pharmaceuticals that require specialized handling and disposal due to their high toxicity.
- U-listed Material: This includes toxic pharmaceutical substances that, while not acutely hazardous, still present significant risks. Examples include certain antidepressants and antibiotics that can adversely affect human health and the environment if improperly managed.
- Non-Creditable Hazardous Material: Pharmaceuticals that are expired, damaged, or otherwise unsuitable for use fall into this category and must be treated as hazardous substances to prevent environmental contamination.
Recent updates indicate that a growing percentage of medical facilities are now managing P-listed and U-listed materials more effectively, reflecting an increased awareness of regulatory requirements. Waste disposal experts emphasize the importance of adhering to these classifications to associated with improper disposal of RCRA pharmaceutical waste. Case studies from Traverse City demonstrate successful execution of disposal protocols that distinguish between P-listed and U-listed materials, showcasing best practices that improve safety and adherence in medical environments. For comprehensive guidelines on managing pharmaceutical debris, refer to the user manuals provided by [Superior Medical Disposal](https://superiorwastedisposal.com), which outline best practices and compliance strategies tailored for healthcare facilities in Michigan, Ohio, and Indiana.
Implement Source Segregation for Effective Waste Management
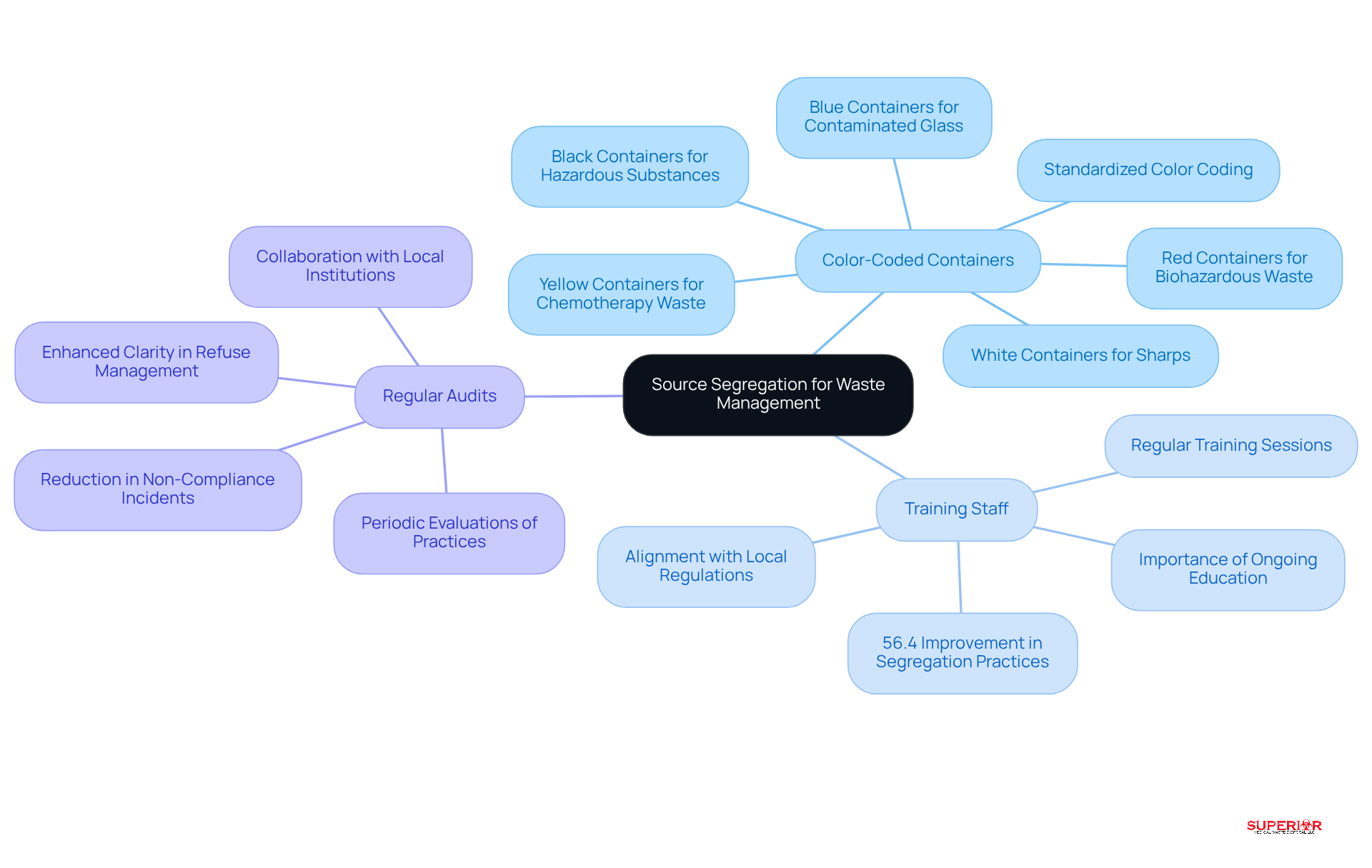
Effective management of pharmaceutical materials, including RCRA pharmaceutical waste, relies heavily on the practice of source segregation. To enhance compliance and operational efficiency, consider implementing the following strategies:
- Color-Coded Containers: Clearly labeled, color-coded containers should be utilized for different types of pharmaceutical waste. For instance, red containers are designated for biohazardous materials, while black containers are suitable for hazardous substances. This system simplifies the disposal process and reduces the risk of contamination.
- Training Staff: Regular training sessions are crucial for ensuring that staff members comprehend the importance of refuse segregation and the correct disposal procedures for various types of pharmaceutical refuse. Statistics indicate that healthcare facilities conducting regular training-preferably at least quarterly-experience a 56.4% improvement in segregation practices among staff. This underscores the importance of ongoing education in fostering a culture of compliance, particularly in light of local regulations such as Michigan’s 90-day removal requirement for biohazard materials, including RCRA pharmaceutical waste.
- Regular Audits: Conducting periodic evaluations of refuse handling practices is vital for ensuring compliance and identifying areas for improvement. Facilities that perform regular audits report enhanced clarity in refuse management and a reduction in non-compliance incidents. For example, our collaboration with Traverse City medical institutions has shown that effective disposal practices can significantly decrease the volume of clinical refuse while ensuring adherence to state regulations.
By adopting these strategies, medical institutions can substantially mitigate the risks associated with improper refuse disposal, particularly regarding RCRA pharmaceutical waste, ensuring compliance with regulatory standards and fostering a safer environment for both personnel and patients.
Develop Customized Waste Management Plans
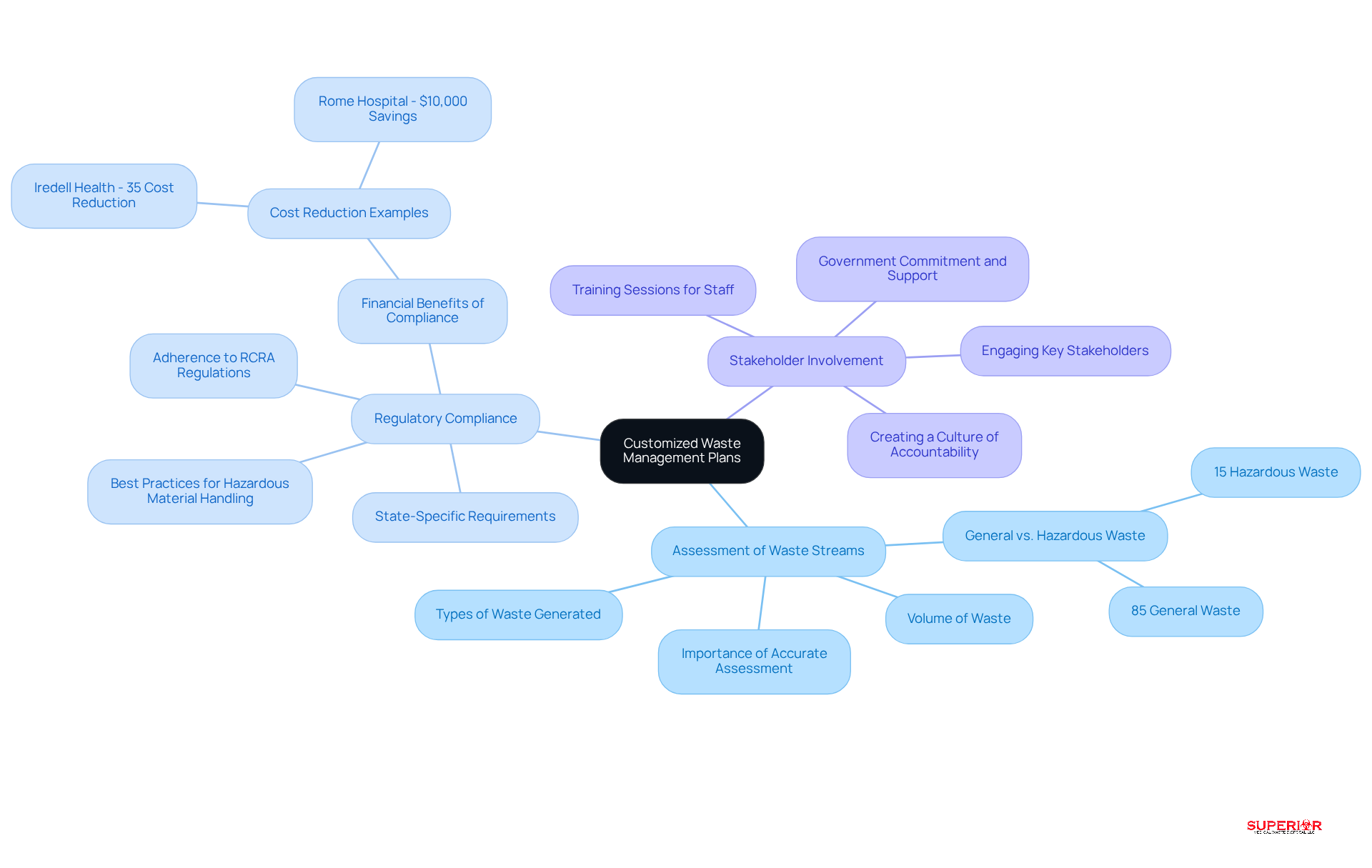
Developing a tailored disposal strategy is essential for healthcare establishments to manage pharmaceutical waste effectively. This strategy should encompass several key components:
- Assessment of Waste Streams: A thorough assessment of the types and volumes of pharmaceutical waste generated is vital. This evaluation informs the creation of a that meets the specific needs of the facility. Notably, around 85% of healthcare waste is classified as general and non-hazardous, while 15% is hazardous. This distinction highlights the necessity of accurate waste stream assessments. The World Health Organization underscores that proper waste segregation is crucial to prevent adverse health outcomes linked to inadequate practices.
- Regulatory compliance necessitates that the disposal plan must adhere to the Resource Conservation and Recovery Act (RCRA) regulations, particularly concerning RCRA pharmaceutical waste and any state-specific requirements. Implementing best practices for hazardous material handling ensures compliance and mitigates potential legal risks. Superior Medical Waste Disposal provides expert guidance and services to assist healthcare facilities in navigating these complex regulations. For instance, Iredell Health achieved a 35% reduction in costs by properly separating and classifying pharmaceutical materials, demonstrating the financial benefits of effective disposal strategies.
- Stakeholder Involvement: Engaging key stakeholders-including staff, leadership, and disposal partners-in the planning process is crucial for fostering buy-in and compliance with the disposal plan. Successful initiatives often feature interactive training sessions that empower staff to advocate for efficient disposal practices. As noted by the World Health Organization, government commitment and support are essential for long-term improvements in waste management. This collaborative approach not only enhances compliance but also cultivates a culture of accountability within the organization.
By developing a tailored waste management plan, healthcare facilities can improve compliance, reduce risks, and enhance overall waste management practices, ultimately contributing to safer and more sustainable healthcare operations.
Conclusion
Effectively managing RCRA pharmaceutical waste is essential for healthcare facilities aiming for compliance and safety. Recognizing the specific regulations that classify certain pharmaceuticals as hazardous enables organizations to establish a framework that meets legal requirements while safeguarding human health and the environment. Proper identification, segregation, and disposal of hazardous pharmaceutical waste are crucial for enhancing operational efficiency and compliance.
This article explores key aspects of effective pharmaceutical waste management, including:
- Identifying hazardous waste types
- Implementing source segregation strategies
- Developing customized waste management plans
Each component is vital for minimizing contamination risks and ensuring adherence to RCRA regulations. Furthermore, emphasizing staff training, conducting regular audits, and involving stakeholders fosters a culture of compliance and accountability within healthcare settings.
In summary, managing RCRA pharmaceutical waste is not only a regulatory obligation but also a moral imperative for healthcare providers. By adopting best practices and staying informed about current trends and compliance guidelines, organizations can significantly mitigate risks associated with hazardous waste. This proactive approach not only protects the environment but also enhances the overall safety and sustainability of healthcare operations. Embracing these strategies will contribute to a healthier future for both patients and communities.
Frequently Asked Questions
What are RCRA regulations regarding pharmaceutical waste?
RCRA regulations govern the management of hazardous materials, including pharmaceuticals, in healthcare settings, classifying certain pharmaceuticals as hazardous based on their potential threat to human health or the environment.
How is hazardous waste defined under RCRA?
Hazardous waste is defined by RCRA as any waste that poses a significant threat to human health or the environment, including pharmaceuticals that are ignitable, corrosive, reactive, or toxic.
Can you provide examples of pharmaceuticals classified as hazardous?
Examples of hazardous pharmaceuticals include chloral hydrate (classified as U034) and epinephrine (classified as P042).
What are the compliance requirements for healthcare establishments?
Healthcare establishments must comply with federal and state regulations, which may require obtaining permits for hazardous material management, maintaining accurate documentation, and retaining shipping manifests and treatment certificates for three to five years.
What are the consequences of failing to comply with RCRA regulations?
Failure to comply with RCRA regulations can result in penalties of up to $25,000 per day for violations.
What training is required for staff handling pharmaceutical waste?
Staff must receive training on RCRA pharmaceutical waste regulations to ensure compliance and safe handling of hazardous materials, with regular updates to training programs as regulations evolve.
Why is understanding RCRA regulations important for healthcare organizations?
Understanding RCRA regulations allows healthcare organizations to implement effective disposal management strategies that prioritize safety, compliance, and environmental responsibility.
