Introduction
Navigating the complex landscape of pharmacy waste disposal presents significant challenges for healthcare facilities, especially as regulations evolve and environmental concerns grow. Understanding the regulatory framework is not merely about compliance; it represents an opportunity for organizations to enhance safety and sustainability in their waste management practices.
How can facilities adopt best practices that not only fulfill legal requirements but also promote public health and environmental protection? This article explores effective strategies for pharmacy waste disposal, providing insights into compliance, safety measures, and the benefits of collaborating with specialized services.
Understand Regulatory Framework for Pharmacy Waste Disposal
is governed by a complex framework of regulations at both federal and state levels, primarily shaped by the and the Drug Enforcement Administration (DEA) regulations. Healthcare facilities must navigate these regulations to avoid significant penalties and ensure safe .
- Classification of Waste: Differentiating between hazardous and non- is crucial. include pharmaceuticals that are toxic, ignitable, corrosive, or reactive, necessitating stringent handling and removal protocols.
- Storage Requirements: Facilities must comply with specific storage guidelines, which include using appropriate containers that are clearly labeled to prevent contamination and ensure safety. Proper ventilation and secure access are essential to associated with unauthorized access.
- : Familiarity with approved is vital. generally require incineration, while unused medications may be handled through , offering a secure method for elimination and decreasing environmental impact.
- : is critical for compliance. Facilities must maintain detailed records of refuse disposal activities, including manifests for , to demonstrate adherence to regulations during inspections. Failure to produce documentation can lead to non-compliance, resulting in penalties that can reach up to $25,000 per day in some states.
By thoroughly understanding these regulations, healthcare facilities can implement effective disposal strategies that prioritize safety and compliance, ultimately protecting public health and the environment.
Develop a Comprehensive Waste Management Plan
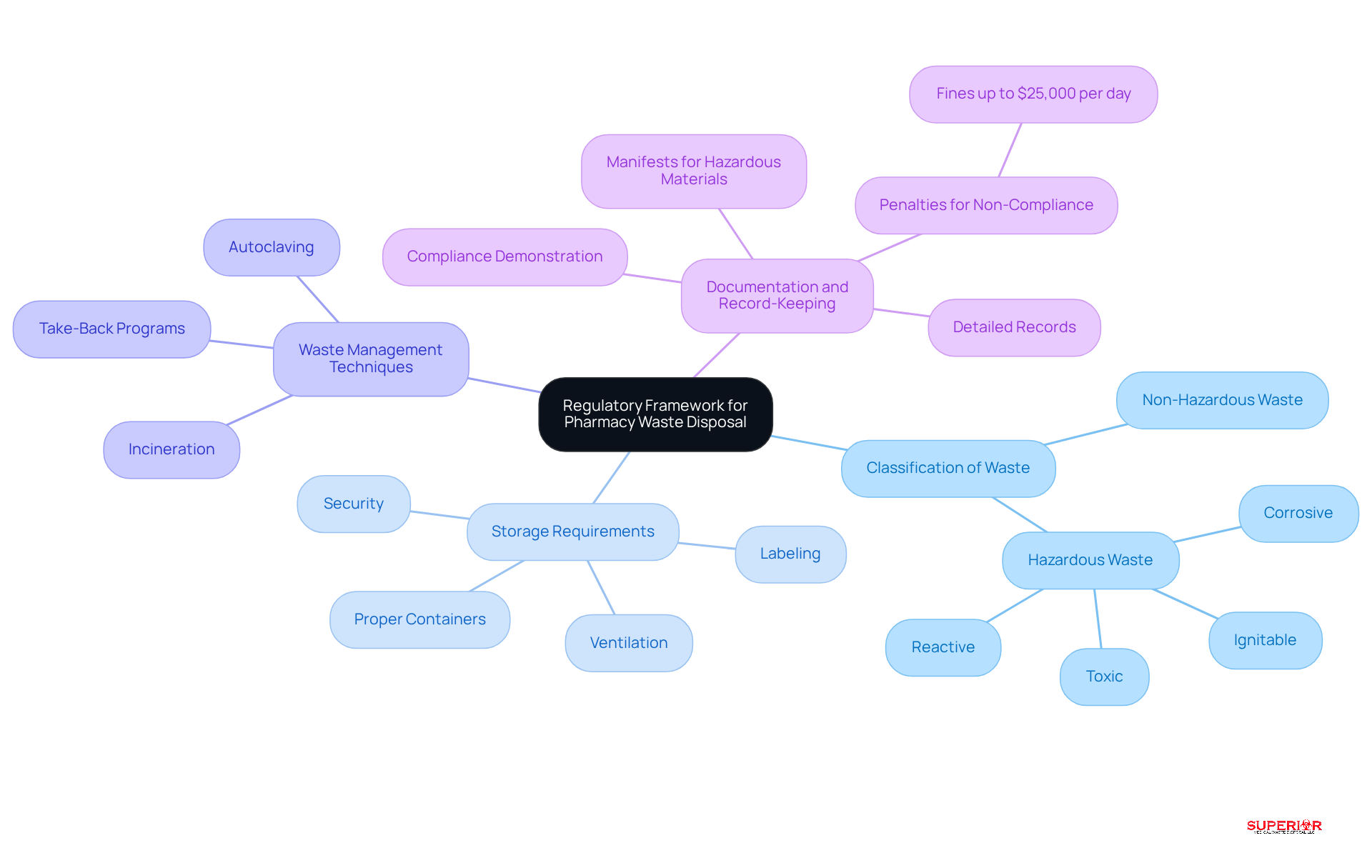
A comprehensive strategy for is essential for the effective management of medicinal byproducts. This plan comprises several key components:
- Assessment of : A detailed evaluation is necessary to identify the types and volumes of pharmaceutical waste generated by the facility. This assessment is crucial for understanding and implementing targeted management strategies.
- Clear protocols for must be established to separate at the point of generation. This practice of helps prevent cross-contamination and ensures compliance with regulatory standards.
- Containerization: Appropriate containers should be utilized for different types of refuse, ensuring they are clearly labeled and compliant with regulations. Proper containerization minimizes the risk of accidental exposure and facilitates safe handling.
- Elimination Procedures: Specific processes for must be outlined, including timelines and methods to be employed. Adhering to these procedures is vital for maintaining safety and regarding . For instance, it is essential to consult local environmental authorities regarding regulations on , as these can vary by state. In Michigan, for example, that must be removed by a licensed medical refuse hauler within 90 days of storage.
- Emergency Response Plan: A comprehensive strategy should be created to address spills or accidents involving pharmaceutical materials. This plan must include training for staff on proper procedures to effectively mitigate risks.
Implementing a thorough strategy not only ensures regulatory adherence but also enhances the safety and efficiency of waste handling operations, ultimately leading to improved health outcomes and environmental sustainability. By addressing the significant , which accounts for over 4% of global CO2 emissions, facilities can demonstrate their commitment to sustainability. At Superior Medical Waste Management, our biohazard material is autoclaved and shredded to reduce volume by 70% before disposal, ensuring compliance and safety in our operations. Our partnerships with leading organizations further bolster our expertise and commitment to best practices in .
Implement Staff Training Programs for Compliance and Safety
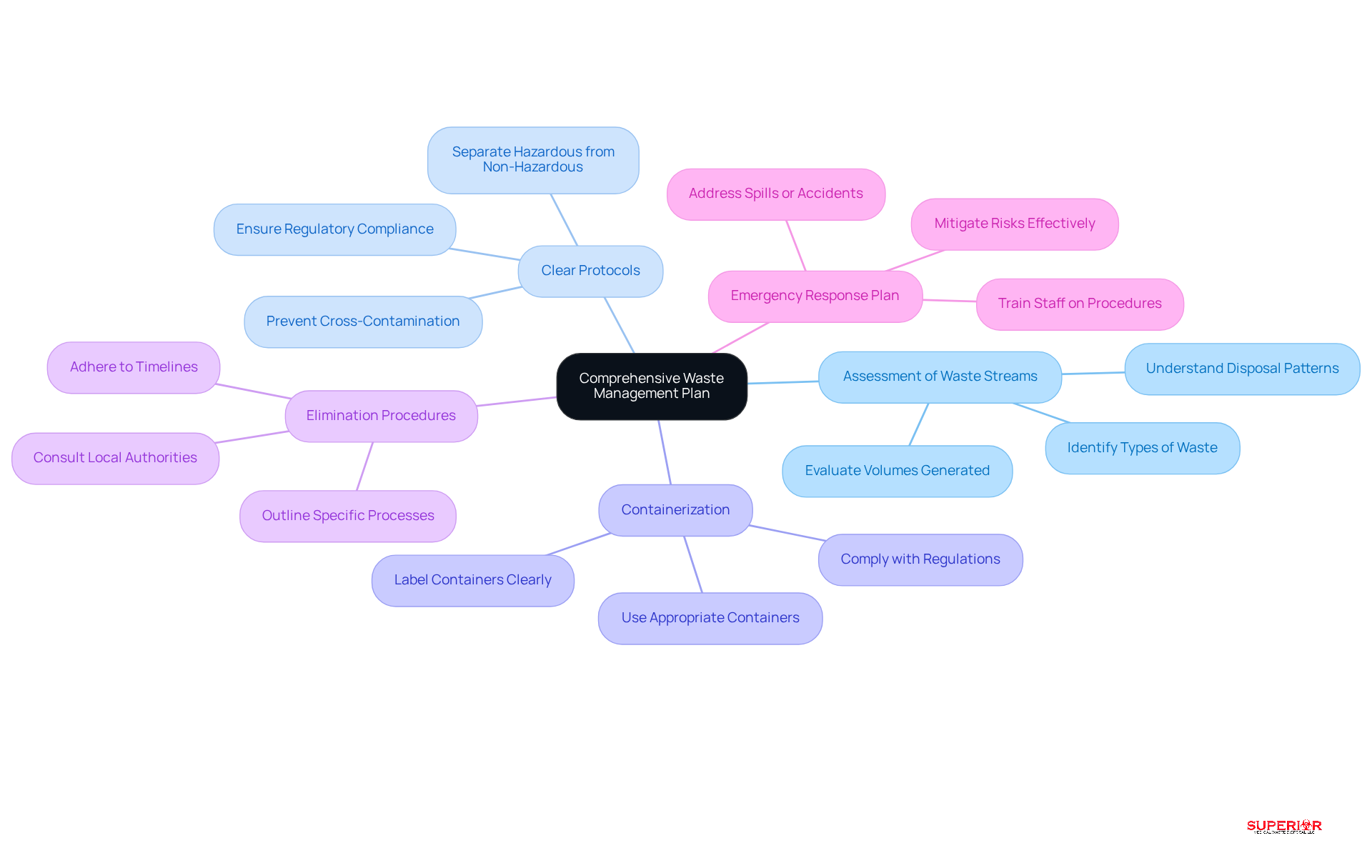
Effective are crucial for ensuring regarding pharmacy management. A successful should include several key components:
- Regular Training Sessions: Consistent training sessions are essential to keep staff updated on the latest regulations and .
- : Practical, hands-on instruction is vital. This approach emphasizes the correct management, separation, and of medicinal refuse, reinforcing theoretical knowledge through real-world application.
- Assessment and Feedback: Implementing assessments allows for gauging staff understanding and providing constructive feedback, which enhances knowledge retention and application.
- : Maintaining detailed records of training sessions, including attendance and topics covered, is necessary to demonstrate compliance during audits and inspections.
- : Encouraging continuous learning and awareness regarding through newsletters, workshops, or online courses ensures that staff remain informed about evolving practices and regulations.
By investing in comprehensive s, healthcare facilities empower their staff to manage safely and effectively, ensuring adherence to regulatory standards and enhancing overall safety.
Partner with Specialized Waste Disposal Services
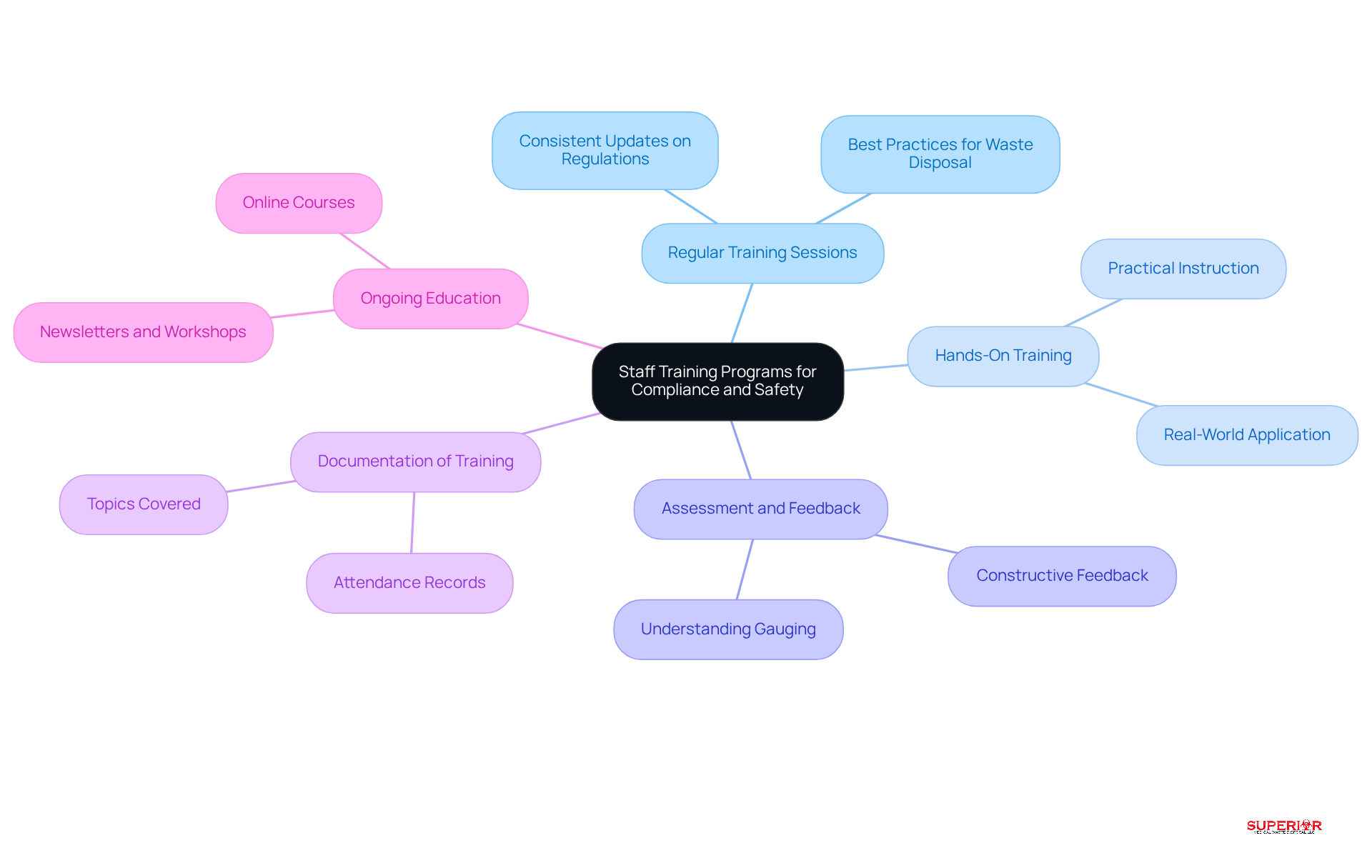
Collaborating with specialized services significantly enhances the efficiency of managing medication byproducts. When selecting a partner, consider the following criteria:
- Expertise and Compliance: Choose a with a proven track record in handling and a thorough understanding of relevant regulations. This related to pharmacy waste disposal, thereby minimizing risks associated with improper waste management.
- Customized Solutions: Look for a partner that offers tailored disposal management solutions to address the specific needs of your facility. and enhance operational efficiency.
- : Verify that the adheres to stringent to protect staff and the environment during waste handling and transportation. A strong commitment to safety is vital for .
- : Opt for a partner that provides clear and structures to prevent unexpected costs. Understanding the financial implications in advance aids in budget oversight and long-term planning.
- Regular Audits and Reporting: Collaborate with a service that conducts routine assessments and offers comprehensive reports on activities. This practice ensures adherence and accountability, enabling healthcare facilities to effectively monitor their refuse handling performance.
By partnering with specialized services for pharmacy waste disposal, healthcare facilities can enhance their refuse handling practices, ensuring safety and compliance while alleviating the burden on internal resources. Statistics show that organizations collaborating with professional experience notable improvements in compliance, further highlighting the value of these partnerships.
Conclusion
Pharmacy waste disposal is not just a regulatory requirement; it is essential for public health and environmental safety. By following best practices in waste management, healthcare facilities can effectively reduce the risks associated with hazardous pharmaceutical waste while ensuring compliance with strict regulations.
This article underscores several key aspects of effective pharmacy waste disposal:
- Understanding the regulatory framework
- Developing comprehensive waste management plans
- Implementing thorough staff training programs
- Collaborating with specialized waste disposal services
Each of these elements is vital in establishing a safe and compliant waste management strategy that safeguards both staff and the environment.
Highlighting the importance of these practices is crucial for the healthcare sector. As regulations evolve and the environmental impact of waste management comes under increasing scrutiny, facilities must prioritize compliance and safety. Engaging in proactive waste disposal strategies not only meets legal obligations but also reflects a commitment to sustainability and public health. By taking decisive action now, healthcare providers can help foster a safer and more environmentally responsible future.
Frequently Asked Questions
What governs pharmaceutical waste management?
Pharmaceutical waste management is governed by a complex framework of regulations at both federal and state levels, primarily shaped by the Resource Conservation and Recovery Act (RCRA) and the Drug Enforcement Administration (DEA) regulations.
Why is it important to classify pharmaceutical waste?
Differentiating between hazardous and non-hazardous pharmaceutical waste is crucial because hazardous materials include pharmaceuticals that are toxic, ignitable, corrosive, or reactive, requiring stringent handling and removal protocols.
What are the storage requirements for pharmaceutical waste?
Facilities must comply with specific storage guidelines, including using appropriate containers that are clearly labeled, ensuring proper ventilation, and providing secure access to mitigate risks associated with unauthorized access.
What waste management techniques should healthcare facilities be familiar with?
Healthcare facilities should be familiar with approved waste management techniques, such as incineration for hazardous materials and take-back programs for unused medications, which provide a secure method for disposal and reduce environmental impact.
Why is documentation and record-keeping important in pharmaceutical waste disposal?
Accurate record-keeping is critical for compliance, as facilities must maintain detailed records of refuse disposal activities, including manifests for hazardous materials, to demonstrate adherence to regulations during inspections.
What are the consequences of failing to maintain proper documentation?
Failure to produce documentation can lead to non-compliance, resulting in penalties that can reach up to $25,000 per day in some states.
List of Sources
- Understand Regulatory Framework for Pharmacy Waste Disposal
- Key Healthcare Regulatory Updates From 2024 and What to Expect in 2025 (https://stericycle.com/en-us/resource-center/blog/key-healthcare-regulatory-updates-from-2024-and-what-to-expect-in-2025)
- Medical Waste Disposal Regulations (2026): A State-by-State Compliance Checklist (https://medprodisposal.com/medical-waste-disposal-regulations-2026-state-by-state-checklist)
- Management of Hazardous Waste Pharmaceuticals | US EPA (https://epa.gov/hwgenerators/management-hazardous-waste-pharmaceuticals)
- RCRA Haz Waste Rules to Watch in 2026 (https://lion.com/lion-news/october-2025/3-rcra-rules-to-watch-in-2026)
- Develop a Comprehensive Waste Management Plan
- remedi Expands Waste Tracking and Reporting to Support Hospital Sustainability and Procurement Teams (https://prnewswire.com/news-releases/remedi-expands-waste-tracking-and-reporting-to-support-hospital-sustainability-and-procurement-teams-302674678.html)
- ghx.com (https://ghx.com/the-healthcare-hub/healthcare-waste-reduction-guide)
- The Future of Waste Management in Hospitals: Trends to Watch (https://danielshealth.com/knowledge-center/future-of-waste-management)
- The Healthcare Waste Challenge: Why Traditional Methods Are Failing America’s Healthcare System – Amlon (https://amlongroup.com/news/the-healthcare-waste-challenge-why-traditional-methods-are-failing-americas-healthcare-system)
- Implement Staff Training Programs for Compliance and Safety
- 30 Inspiring Learning and Development Quotes (https://intellum.com/resources/blog/learning-and-development-quotes)
- 2026 predictions: Managing the medical waste challenge in home care (https://mcknightshomecare.com/2026-predictions-managing-the-medical-waste-challenge-in-home-care)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC12598056)
- Effectiveness of intensive healthcare waste management training model among health professionals at teaching hospitals of Pakistan: a quasi-experimental study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4353471)
- Partner with Specialized Waste Disposal Services
- 10 Reasons to Invest in Biomedical Waste Management Services (https://uffizio.com/blog/biomedical-waste-management-services)
- Benefits of Proper Medical Waste Segregation (https://stericycle.com/en-us/resource-center/blog/benefits-of-proper-medical-waste-segregation)
- Bio-MED Regulated Waste Solutions (https://getbiomed.com/medical-waste/the-benefits-of-working-with-a-medical-waste-management-company-vs-a-medical-waste-hauler-or-broker)
