Introduction
Understanding the complexities of pharmaceutical waste management is essential for healthcare administrators responsible for ensuring compliance and protecting public health. This guide provides a thorough overview of the regulations, types of waste, and best practices necessary for developing an effective disposal program. With laws continually evolving and state-specific guidelines differing, administrators face the challenge of navigating these complexities to implement a sustainable and compliant waste management strategy.
Understand Pharmaceutical Waste Management Regulations
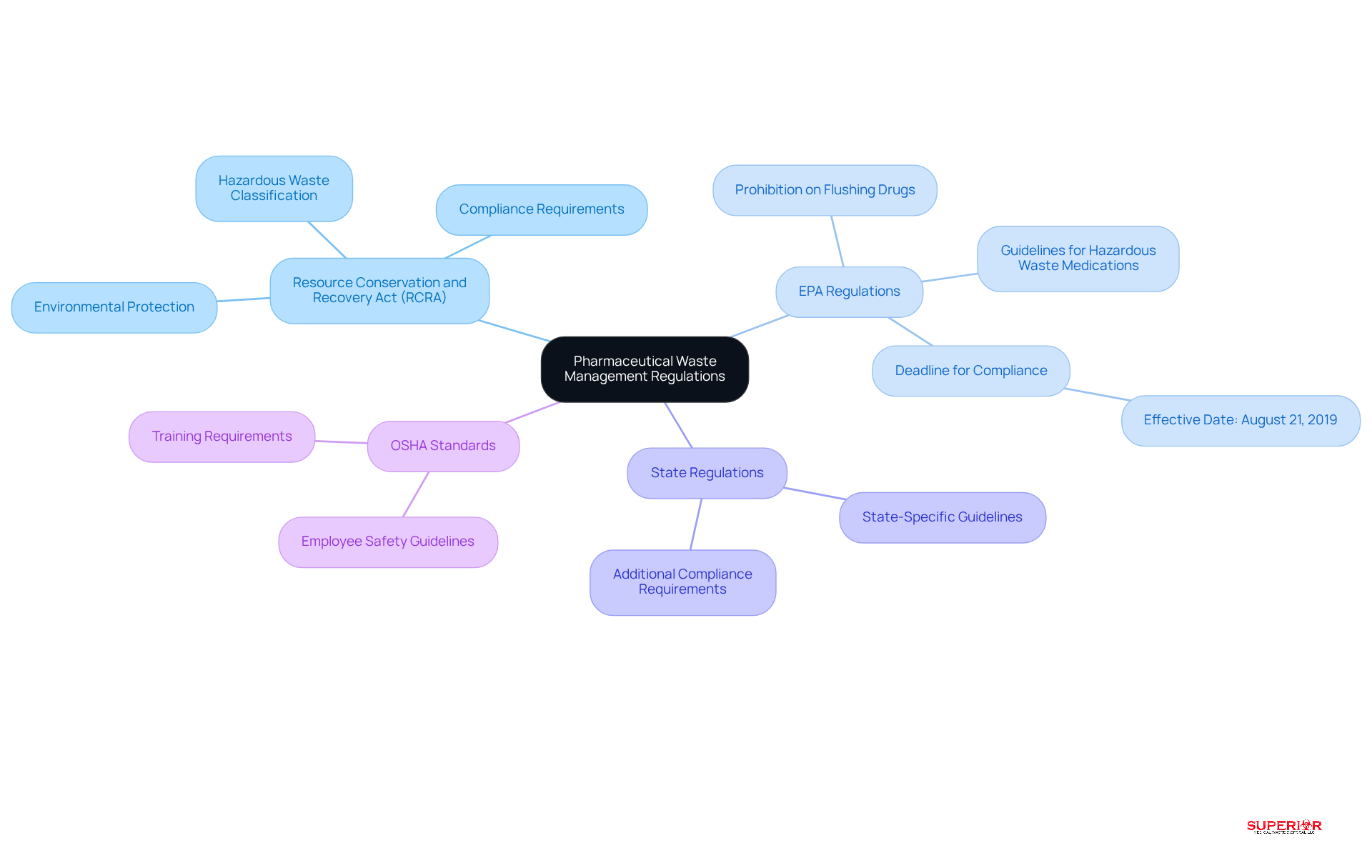
To effectively manage pharmaceutical waste, administrators must understand key regulations that govern disposal practices:
- Resource Conservation and Recovery Act (RCRA): This federal legislation regulates the disposal of hazardous materials, including medications. Facilities must assess whether their waste is classified as hazardous under RCRA, which is crucial for managing pharmaceutical waste, compliance, and environmental protection.
- EPA Regulations: The Environmental Protection Agency (EPA) has established specific guidelines for managing hazardous waste medications. Notably, there is a prohibition against flushing certain drugs down toilets or sinks as part of managing pharmaceutical waste. This regulation aims to prevent the contamination of water sources and ecosystems.
- State Regulations: Each state may impose additional regulations that complement federal laws. It is essential for administrators to consult state-specific guidelines to ensure comprehensive compliance and avoid potential penalties.
- OSHA Standards: The Occupational Safety and Health Administration (OSHA) sets forth guidelines that healthcare facilities must follow to protect employees handling pharmaceutical materials. Adhering to OSHA standards is vital for maintaining a safe workplace.
By thoroughly understanding these regulations, administrators can develop a compliant disposal management program focused on managing pharmaceutical waste, which minimizes legal risks and enhances safety within their facilities. This proactive approach not only safeguards public health but also aligns with sustainable waste management practices, ultimately contributing to a healthier environment.
Identify Types of Pharmaceutical Waste in Your Facility
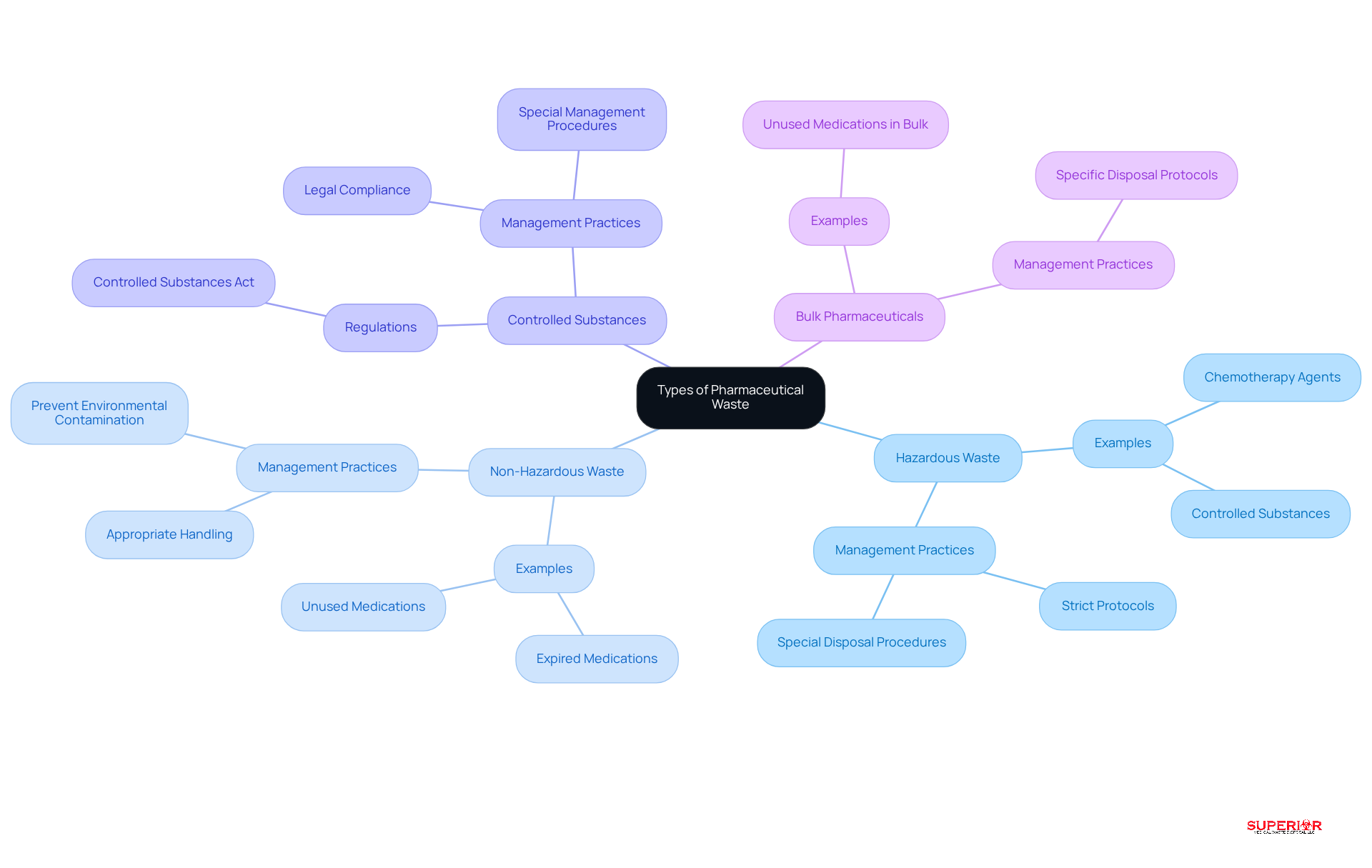
Pharmaceutical waste can be categorized into several distinct types, each requiring specific management practices:
- Hazardous Waste: This category includes substances that pose significant risks to human health or the environment, such as chemotherapy agents and certain controlled substances. Approximately 15% of healthcare waste is categorized as hazardous, which necessitates strict protocols for managing pharmaceutical waste.
- Non-Hazardous Waste: This encompasses expired or unused medications that do not meet hazardous criteria. While these items are less hazardous, managing still requires appropriate handling to prevent environmental contamination.
- Controlled Substances: These drugs are regulated under the Controlled Substances Act and necessitate special management and disposal procedures to prevent misuse and ensure compliance with legal standards.
- Bulk Pharmaceuticals: This includes unused or expired medications that are not packaged in individual doses. Their disposal may involve different methods, often requiring specific protocols for managing pharmaceutical waste to mitigate risks.
To effectively recognize these types of refuse, conduct a thorough inventory of all medications used in your facility. This inventory should categorize items based on their characteristics and regulatory requirements, aligning with current best practices for 2025. Ongoing training and awareness among staff regarding managing pharmaceutical waste and the differences between hazardous and non-hazardous materials are essential for upholding regulations and protecting public health. [Superior Medical Disposal](https://superiorwastedisposal.com) offers expert handling services for hazardous materials, including OSHA compliance training for staff to ensure legal adherence and safe practices.
Develop a Compliant Pharmaceutical Waste Disposal Program
To establish a compliant pharmaceutical waste disposal program, follow these essential steps:
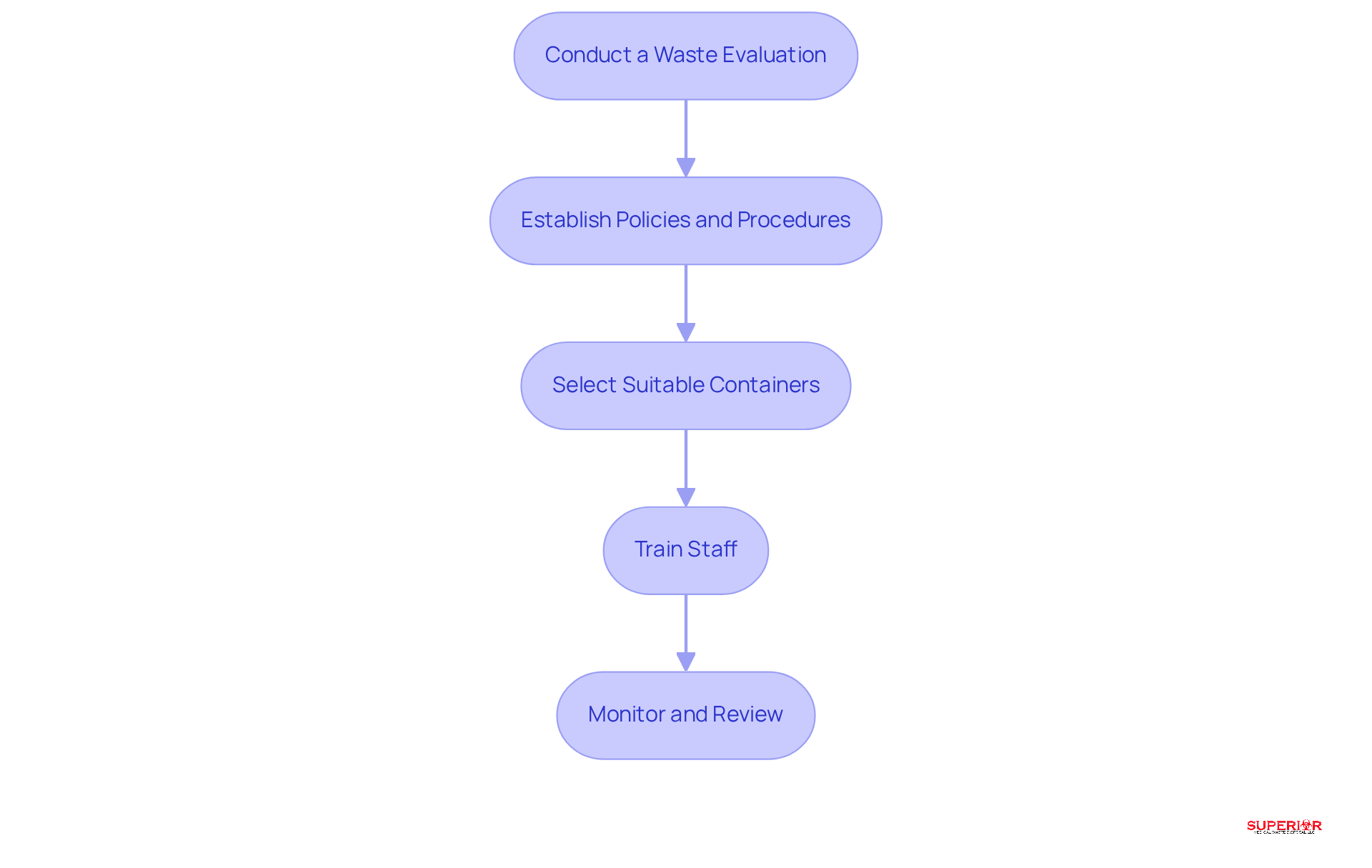
- Conduct a Waste Evaluation: Begin by assessing the types and quantities of pharmaceutical waste generated within your facility. This evaluation is crucial for understanding waste flows and identifying areas for improvement. Healthcare managers emphasize that effective waste evaluation methods can significantly enhance compliance and operational efficiency.
- Establish Policies and Procedures: Develop comprehensive guidelines that clearly outline the management of drug disposal, including collection, storage, and disposal methods. These policies should align with current regulations and best practices for managing pharmaceutical waste to ensure compliance.
- Select Suitable Containers: Utilize clearly marked, leak-proof containers designated for various types of pharmaceutical waste. This practice prevents cross-contamination and ensures safe handling throughout the waste management process.
- Train Staff: Implement for all staff members regarding the proper handling and disposal of pharmaceutical materials. Highlight the importance of compliance and the critical role each employee plays in managing pharmaceutical waste to uphold safety standards.
- Monitor and Review: Regularly evaluate and update the waste management program to stay in line with evolving regulations and best practices. Continuous oversight ensures that your facility remains compliant and can adapt to any changes in medication disposal strategies.
By adhering to these steps, healthcare facilities can establish a robust medication management program focused on managing pharmaceutical waste, which not only meets regulatory standards but also promotes environmental sustainability and public health.
Implement Best Practices for Waste Segregation and Disposal
To ensure effective pharmaceutical waste management, it is crucial to implement the following best practices:
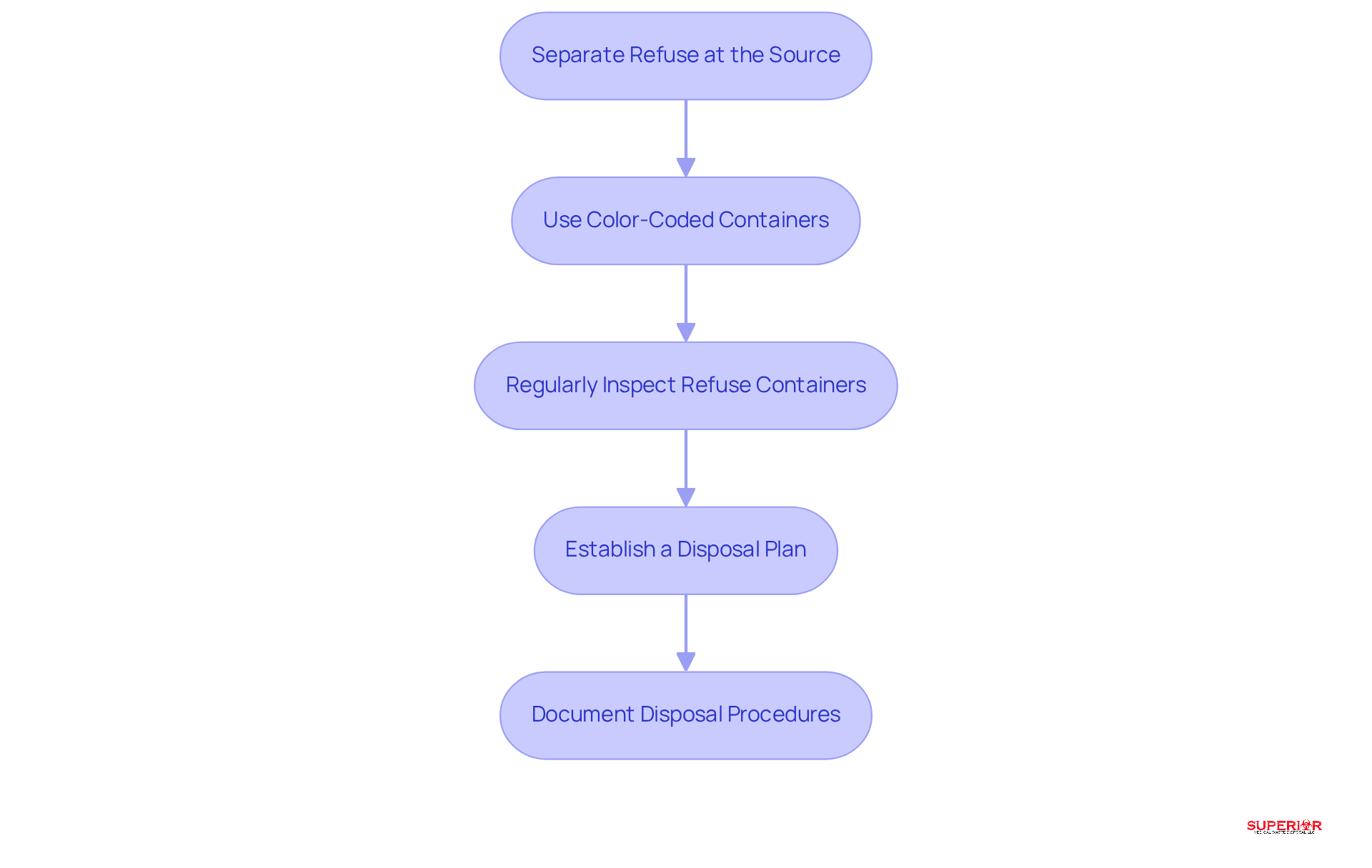
- Separate Refuse at the Source: Educate personnel to immediately divide medical refuse into designated containers upon disposal. This practice is essential to avoid contamination. It is important to recognize that the disposal of medicines is categorized differently from biohazard materials, which is vital for compliance.
- Use Color-Coded Containers: Implement a – red for hazardous materials and black for non-hazardous – to simplify identification and enhance compliance.
- Regularly Inspect Refuse Containers: Conduct routine inspections of refuse containers to ensure they are correctly labeled and not overfilled. This practice aligns with state regulations regarding the storage durations for medical materials.
- Establish a Disposal Plan: Create a timetable for the regular collection and elimination of medical refuse to prevent accumulation and ensure timely removal. Superior Medical Waste Disposal offers reliable handling and removal services tailored for healthcare establishments.
- Document Disposal Procedures: Maintain records of all pharmaceutical waste disposal activities, including dates, quantities, and methods used, to demonstrate compliance during inspections. Additionally, consider utilizing Superior Medical Waste Disposal’s OSHA compliance training services to keep your staff informed about legal requirements.
Conclusion
In conclusion, understanding and managing pharmaceutical waste is essential for healthcare administrators who must ensure compliance and protect public health. This article presents a comprehensive approach to navigating the complexities of pharmaceutical waste management, highlighting the necessity of adhering to regulatory standards while implementing effective disposal practices.
Key points include:
- The importance of grasping various regulations, such as RCRA, EPA guidelines, and state-specific laws, which dictate the handling of pharmaceutical waste.
- The categorization of pharmaceutical waste into hazardous, non-hazardous, controlled substances, and bulk pharmaceuticals, each requiring tailored management strategies.
- Establishing a compliant disposal program involves conducting waste evaluations, creating clear policies, training staff, and continuously monitoring practices to adapt to evolving regulations.
The significance of proper pharmaceutical waste management extends beyond legal compliance; it is vital for safeguarding environmental health and public safety. By prioritizing waste segregation and adhering to best practices, healthcare facilities can minimize risks associated with pharmaceutical waste. This proactive approach not only fulfills regulatory obligations but also contributes to a sustainable future. Administrators are encouraged to implement the strategies outlined to ensure their facilities remain compliant and responsible stewards of environmental health.
Frequently Asked Questions
What is the Resource Conservation and Recovery Act (RCRA)?
The Resource Conservation and Recovery Act (RCRA) is a federal legislation that regulates the disposal of hazardous materials, including medications. It is crucial for facilities to assess whether their waste is classified as hazardous under RCRA for effective pharmaceutical waste management and compliance.
What guidelines does the EPA provide for managing pharmaceutical waste?
The Environmental Protection Agency (EPA) has established specific guidelines for managing hazardous waste medications, including a prohibition against flushing certain drugs down toilets or sinks. This regulation aims to prevent the contamination of water sources and ecosystems.
Are there additional regulations at the state level for pharmaceutical waste management?
Yes, each state may impose additional regulations that complement federal laws. It is essential for administrators to consult state-specific guidelines to ensure comprehensive compliance and avoid potential penalties.
What role does OSHA play in pharmaceutical waste management?
The Occupational Safety and Health Administration (OSHA) sets forth guidelines that healthcare facilities must follow to protect employees handling pharmaceutical materials. Adhering to OSHA standards is vital for maintaining a safe workplace.
Why is it important for administrators to understand pharmaceutical waste management regulations?
Understanding these regulations allows administrators to develop a compliant disposal management program focused on managing pharmaceutical waste. This minimizes legal risks, enhances safety within facilities, safeguards public health, and contributes to sustainable waste management practices.
