Introduction
Navigating the complexities of pharmaceutical hazardous waste management is essential for healthcare facilities aiming to comply with stringent regulations and protect public health. As guidelines from the EPA evolve and the risks associated with improper disposal increase, organizations must implement effective strategies to manage their waste responsibly.
How can healthcare providers ensure compliance while minimizing environmental impact and safeguarding their communities?
Understand Pharmaceutical Hazardous Waste Classifications
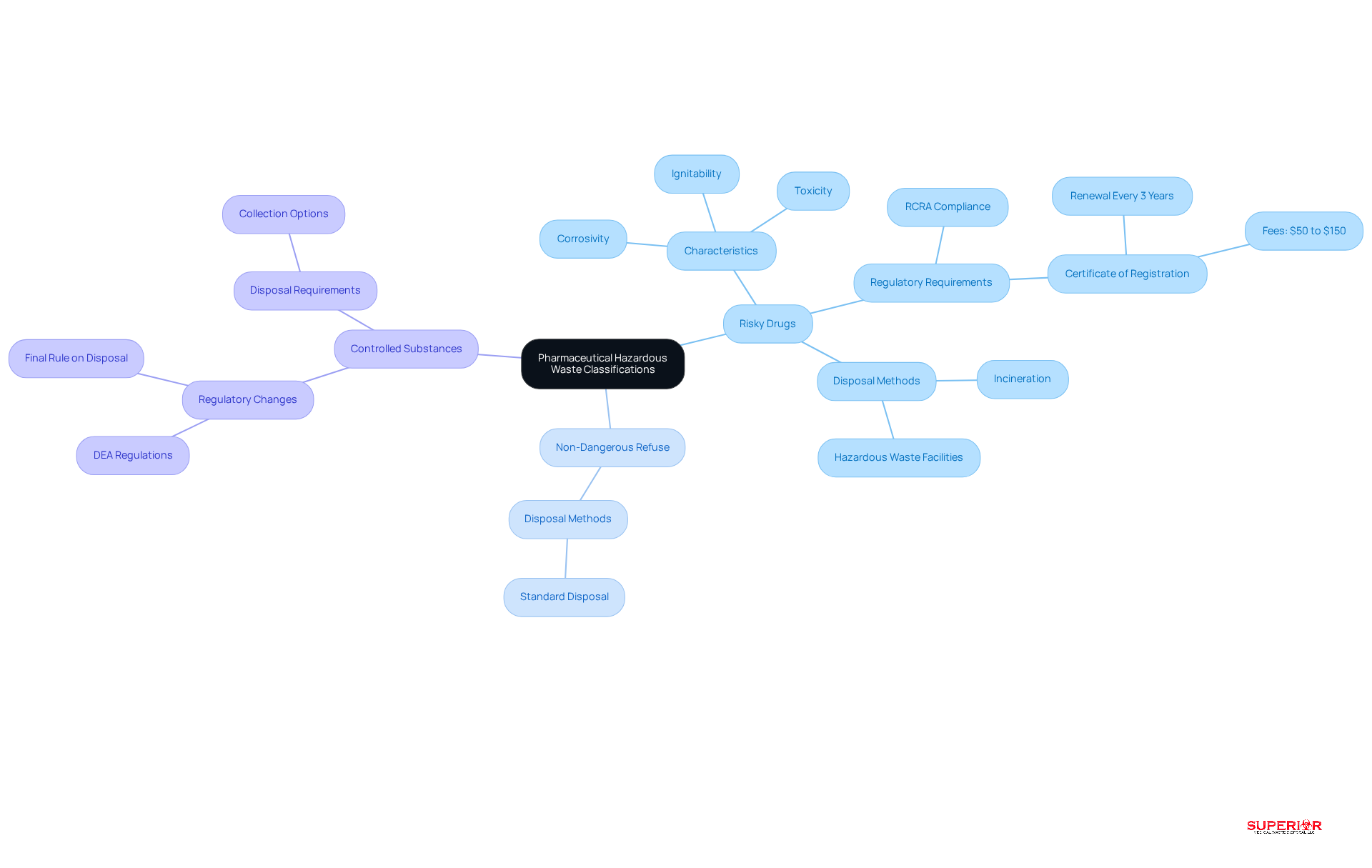
Medical dangerous refuse can be categorized into several groups:
- Risky drugs
- Non-dangerous refuse
- Controlled substances
According to the EPA, hazardous waste includes materials that exhibit one or more hazardous characteristics, such as ignitability, corrosivity, or toxicity, or are specifically listed as hazardous. Understanding these classifications is vital for compliance with the RCRA and other regulations.
Establishments must ensure they possess a valid permit, necessary for any site producing medical byproducts. This certificate must be renewed every three years, with fees ranging from $50 to $150 based on the type of establishment. Furthermore, all facilities are legally obligated to have their sharps and hazardous waste removed every 90 days or less, regardless of the volume generated.
Consistently assessing refuse streams and educating personnel to identify classifications is crucial to avoid fines and potential legal consequences. For instance, controlled substances must be treated as hazardous waste, while non-dangerous drugs can typically be discarded through standard disposal methods.
The new regulation took effect on August 21, 2019, underscoring the importance of compliance with these guidelines. As stated by the EPA, “No dangerous medication materials can be discarded down the drain anywhere in the US,” which emphasizes the necessity of appropriate disposal methods. Additionally, healthcare organizations operating in federally administered states must remain aware of specific regulatory changes that apply to their operations.
Comply with Regulatory Standards for Waste Disposal
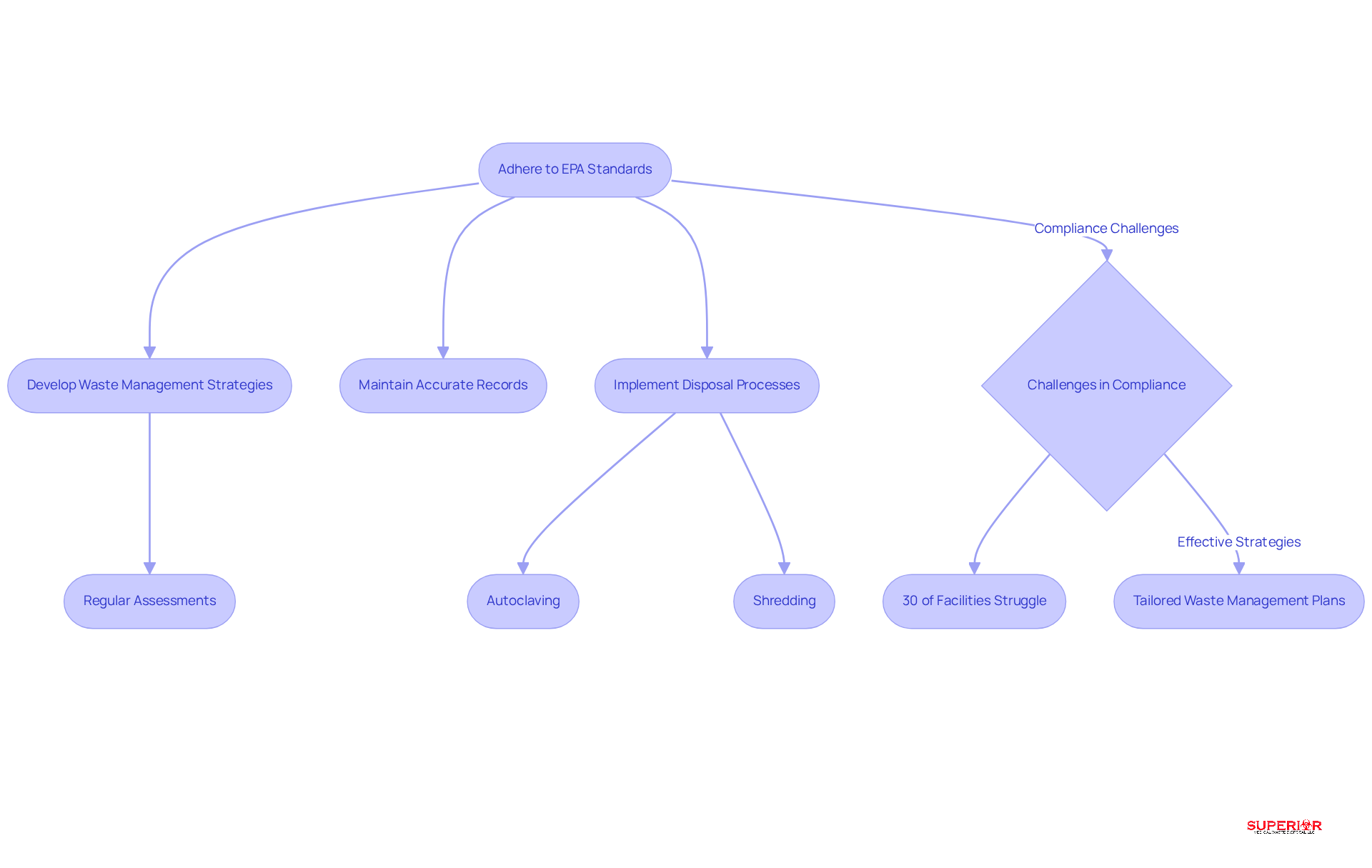
Healthcare establishments must adhere to stringent regulatory standards when managing pharmaceutical waste, particularly those outlined by the EPA under the Resource Conservation and Recovery Act. Recent updates to these regulations mandate that all pharmaceutical waste be disposed of in ways that mitigate environmental contamination risks. To comply with these evolving standards, healthcare facilities should develop waste management plans that incorporate regular assessments and updates.
The organization enhances its expertise and compliance in waste management through a robust network of strategic partners, including Henry Ford Health and Trinity Health. This collaboration provides healthcare organizations with essential support to navigate these regulations effectively.
Moreover, maintaining accurate records of disposal activities such as manifests and training documentation is crucial for demonstrating compliance during inspections. Statistics indicate that approximately 30% of healthcare establishments encounter difficulties in meeting EPA disposal standards for pharmaceutical waste, highlighting the need for robust management practices and continuous education regarding regulatory changes. For instance, case studies reveal that facilities implementing training programs, like those offered by industry leaders, have significantly improved their compliance rates, highlighting the effectiveness of proactive management strategies.
Superior’s waste management strategy includes autoclaving, which involves steam heating materials at high temperatures to eliminate potential biological threats, followed by shredding to reduce volume by 70%. This approach exemplifies best practices for regulatory compliance and efficient waste disposal.
Implement Effective Segregation and Containment Practices
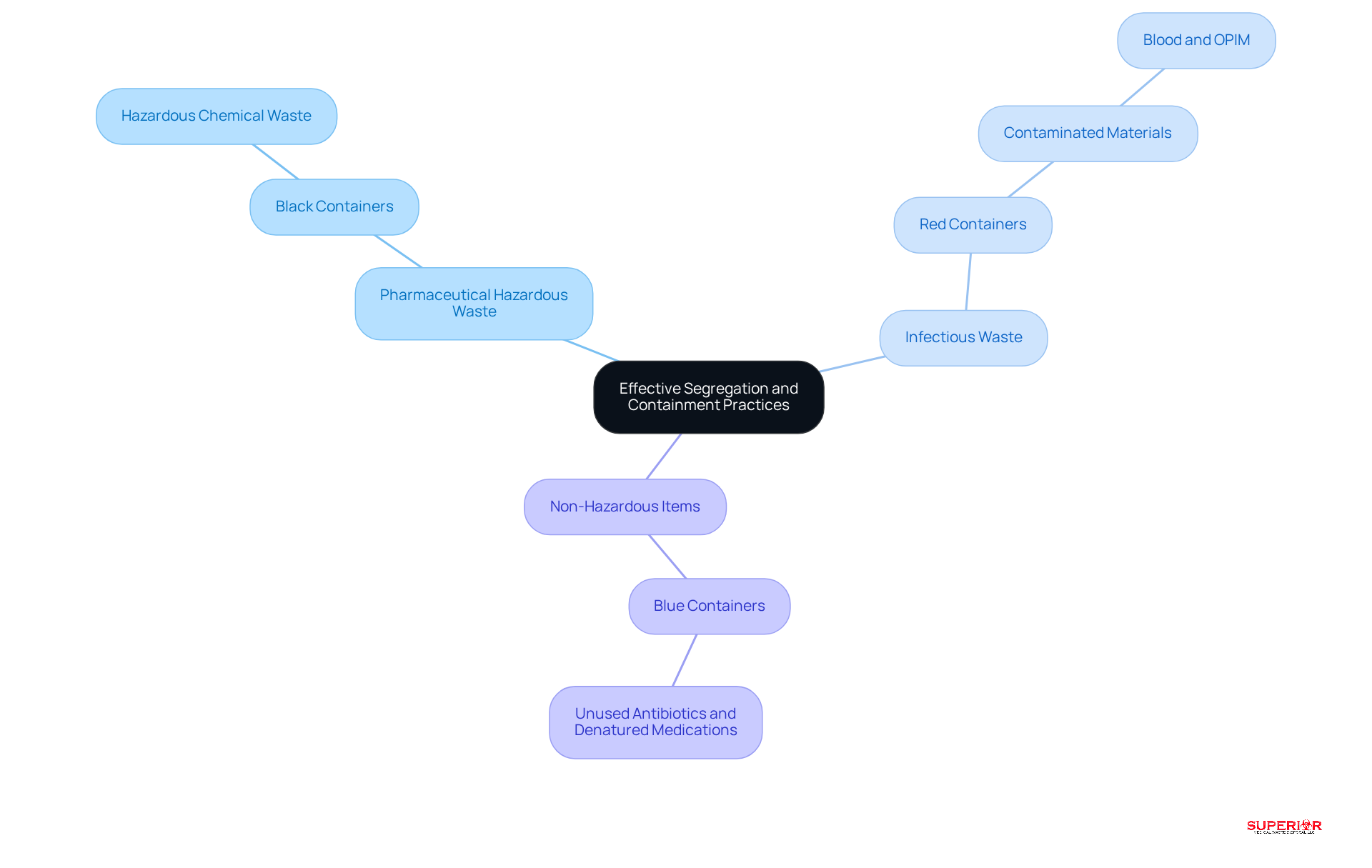
Efficient separation of medicinal refuse is crucial for waste management. A color-coded system allows facilities to clearly differentiate between hazardous waste and other types of waste. For instance:
- Black containers are designated for pharmaceutical waste.
- Red containers are used for materials contaminated with blood or other potentially infectious substances.
- Blue containers are typically reserved for non-hazardous items like unused antibiotics and denatured medications.
Research shows that around 70% of healthcare facilities have implemented color-coded systems to optimize disposal management processes for hazardous materials, as noted in the EPA’s 2022 guide for waste management.
Training staff to segregate waste at the point of generation is vital for preventing contamination and ensuring adherence to regulations. Regular audits can identify areas for improvement, thereby enhancing overall operational efficiency. Additionally, utilizing containers that are appropriate for the specific type of material is essential to prevent leaks and spills during storage and transport. This proactive strategy not only protects public health but also aligns with established practices in waste management.
Train Staff on Safe Pharmaceutical Waste Management
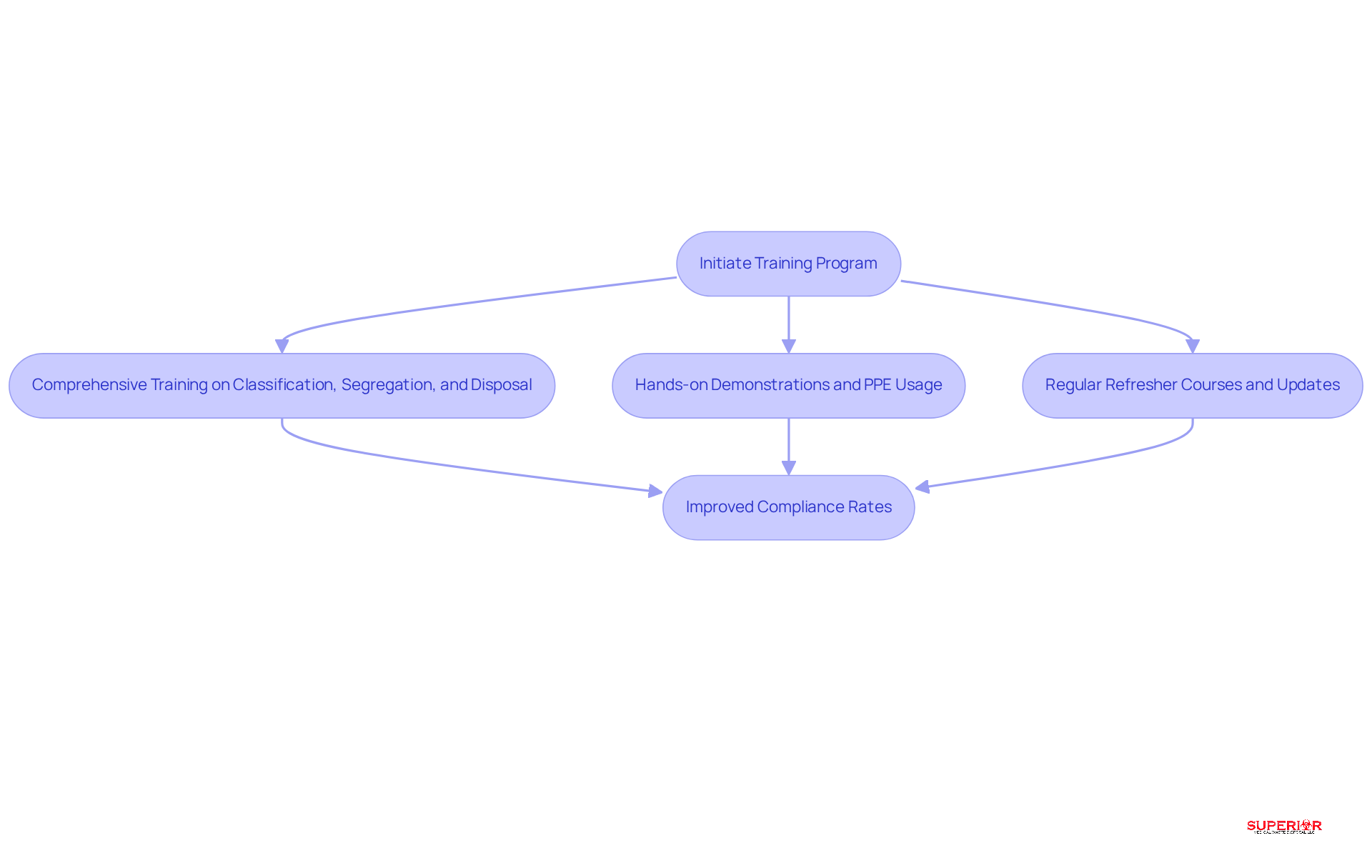
Training personnel on waste management is crucial for ensuring compliance and safety within healthcare facilities. All staff involved in handling hazardous waste must undergo training, a service provided by Superior Medical Disposal. This training highlights the dangers associated with various types of waste and outlines the best practices, emphasizing the importance of using compliant containers for removal.
Regular refresher courses and updates on regulatory changes are essential for keeping staff informed and compliant. Implementing a training program that includes hands-on practice can significantly enhance learning outcomes. Superior Medical Waste Disposal also offers resources to ensure that staff remain legally compliant.
Research indicates that effective training can lead to improved compliance, with some facilities reporting increases from 66% to 90% in adherence to safety protocols. By prioritizing training, healthcare facilities can greatly reduce the risks associated with hazardous waste, ultimately safeguarding both public health and the environment.
Conclusion
Understanding and managing pharmaceutical hazardous waste is essential for healthcare facilities. This responsibility ensures compliance, protects public health, and minimizes environmental impact. By implementing effective practices in waste classification, regulatory compliance, segregation, and staff training, organizations can significantly enhance their waste management processes.
The article emphasizes the importance of recognizing various categories of pharmaceutical waste and adhering to the latest regulations set forth by the EPA. Facilities must develop comprehensive waste management strategies that include regular assessments and clear segregation practices. Furthermore, training staff on safe handling and disposal methods is crucial for maintaining compliance and fostering a culture of safety within the organization.
Ultimately, the responsibility for managing pharmaceutical hazardous waste rests with healthcare providers. By prioritizing adherence to regulations and investing in staff education, facilities can improve compliance rates and contribute to a healthier environment. Commitment to effective waste management practices is not merely a legal obligation; it is a vital step toward safeguarding community health and ensuring sustainable healthcare operations.
Frequently Asked Questions
What are the main categories of pharmaceutical hazardous waste?
Pharmaceutical hazardous waste can be categorized into three groups: risky drugs, non-dangerous refuse, and controlled substances.
What defines pharmaceutical hazardous waste according to the EPA?
Pharmaceutical hazardous waste includes materials that exhibit hazardous characteristics such as ignitability, corrosivity, or toxicity, or are specifically listed as hazardous.
Why is it important to understand pharmaceutical hazardous waste classifications?
Understanding these classifications is vital for compliance with the Resource Conservation and Recovery Act (RCRA) and other regulations to avoid improper disposal and potential legal consequences.
What is required for establishments that produce medical byproducts?
Establishments must possess a valid Certificate of Registration, which must be renewed every three years, with fees ranging from $50 to $150 based on the type of establishment.
How often must sharps and biohazard materials be removed from medical refuse-producing facilities?
All medical refuse-producing facilities are legally obligated to have their sharps and biohazard materials removed every 90 days or less, regardless of the volume generated.
What should facilities do to avoid improper disposal of pharmaceutical hazardous waste?
Facilities should consistently assess refuse streams and educate personnel to identify pharmaceutical hazardous waste classifications.
What are some examples of medications that must be treated as pharmaceutical hazardous waste?
Medications that are flammable, corrosive, or toxic must be treated as pharmaceutical hazardous waste.
What is the EPA’s regulation regarding the disposal of dangerous medication materials?
The EPA states that ‘No dangerous medication materials can be discarded down the drain anywhere in the US,’ emphasizing the necessity of appropriate disposal methods.
What should healthcare organizations in federally administered states be aware of?
Healthcare organizations must remain aware of specific regulatory changes that apply to their operations regarding pharmaceutical hazardous waste disposal.
List of Sources
- Understand Pharmaceutical Hazardous Waste Classifications
- EPA Addresses Challenges in Managing Hazardous Waste Pharmaceuticals | AHE (https://ahe.org/ahe-epa-addresses-challenges-managing-hazardous-waste-pharmaceuticals)
- Industry News (https://pharmecology.com/News/Recent)
- Hazardous Waste Statistics to Know in 2026 | HWH Environmental (https://hwhenvironmental.com/facts-and-statistics-about-waste)
- EPA Overhauls Rules for Pharmaceutical Wastes That Qualify as RCRA Hazardous Wastes (https://bdlaw.com/publications/epa-overhauls-rules-for-pharmaceutical-wastes-that-qualify-as-rcra-hazardous-wastes)
- Management of Hazardous Waste Pharmaceuticals | US EPA (https://epa.gov/hwgenerators/management-hazardous-waste-pharmaceuticals)
- Comply with Regulatory Standards for Waste Disposal
- Key Healthcare Regulatory Updates From 2024 and What to Expect in 2025 (https://stericycle.com/en-us/resource-center/blog/key-healthcare-regulatory-updates-from-2024-and-what-to-expect-in-2025)
- Pharmaceutical Industry, Environmental Protection Agency (EPA), Health Care Providers | JD Supra (https://jdsupra.com/topics/pharmaceutical-industry/environmental-protection-agency-epa/health-care-providers)
- Pharma Rule | US EPA (https://epa.gov/ia/pharma-rule)
- New Subpart P Rule: What the 2025 Hazardous Waste Pharmaceuticals Regulations Mean for Your Facility – Waste Medic (https://wastemedic.com/2025/07/15/new-subpart-p-rule-what-the-2025-hazardous-waste-pharmaceuticals-regulations-mean-for-your-facility)
- Industry News (https://pharmecology.com/News/Recent)
- Implement Effective Segregation and Containment Practices
- wastetodaymagazine.com (https://wastetodaymagazine.com/news/epa-medical-waste-hazardous-guide-disposal)
- As hospital systems begin to grapple with medical waste, environmentalists call for tighter regulations (https://northcarolinahealthnews.org/2023/10/27/as-hospital-systems-begin-to-grapple-with-medical-waste-environmentalists-call-for-tighter-regulations)
- Medical Waste Color Coding in 2025: A Fresh Guide to Safe & Compliant Disposal – Waste Medic (https://wastemedic.com/2025/06/07/medical-waste-color-coding-in-2025-a-fresh-guide-to-safe-compliant-disposal)
- Biomedical Waste Color Coding Guide: OSHA & WHO Standards (https://sdmedwaste.com/post/biomedical-waste-color-coding-guide)
- Biomedical Waste Solutions For 2026 | MedSharps (https://medsharps.com/blog/biomedical-waste-solutions-for-2026)
- Train Staff on Safe Pharmaceutical Waste Management
- How countries in the Americas are strengthening their capacity for safe medical waste management (https://paho.org/en/news/27-10-2025-how-countries-americas-are-strengthening-their-capacity-safe-medical-waste)
- cegh.net (https://cegh.net/article/S2213-3984(24)00022-8/fulltext)
- Healthcare Waste Management training now being rolled out to the Region | Pacific Environment (https://sprep.org/news/healthcare-waste-management-training-now-being-rolled-out-to-the-region)
- bmjopenquality.bmj.com (https://bmjopenquality.bmj.com/content/13/3/e002793)
- Pharmaceutical Waste Disposal (https://getbiomed.com/hazardous-waste/the-importance-of-staff-training-in-hazardous-waste-management)
