Introduction
Managing controlled substances in healthcare settings is not merely a regulatory obligation; it is a vital aspect of public safety. As scrutiny on compliance intensifies and the risks associated with improper disposal become more apparent, healthcare facilities must adopt best practices that ensure legal adherence and environmental protection.
Healthcare providers encounter numerous challenges in navigating the complex landscape of controlled substance regulations. To effectively implement safe disposal procedures and mitigate risks, it is essential to explore strategies that emphasize compliance and the potential consequences of negligence.
This article examines key approaches for the responsible disposal of controlled substances, underscoring the importance of adherence to regulations and the implications of failing to do so.
Understand Controlled Substances and Their Regulations
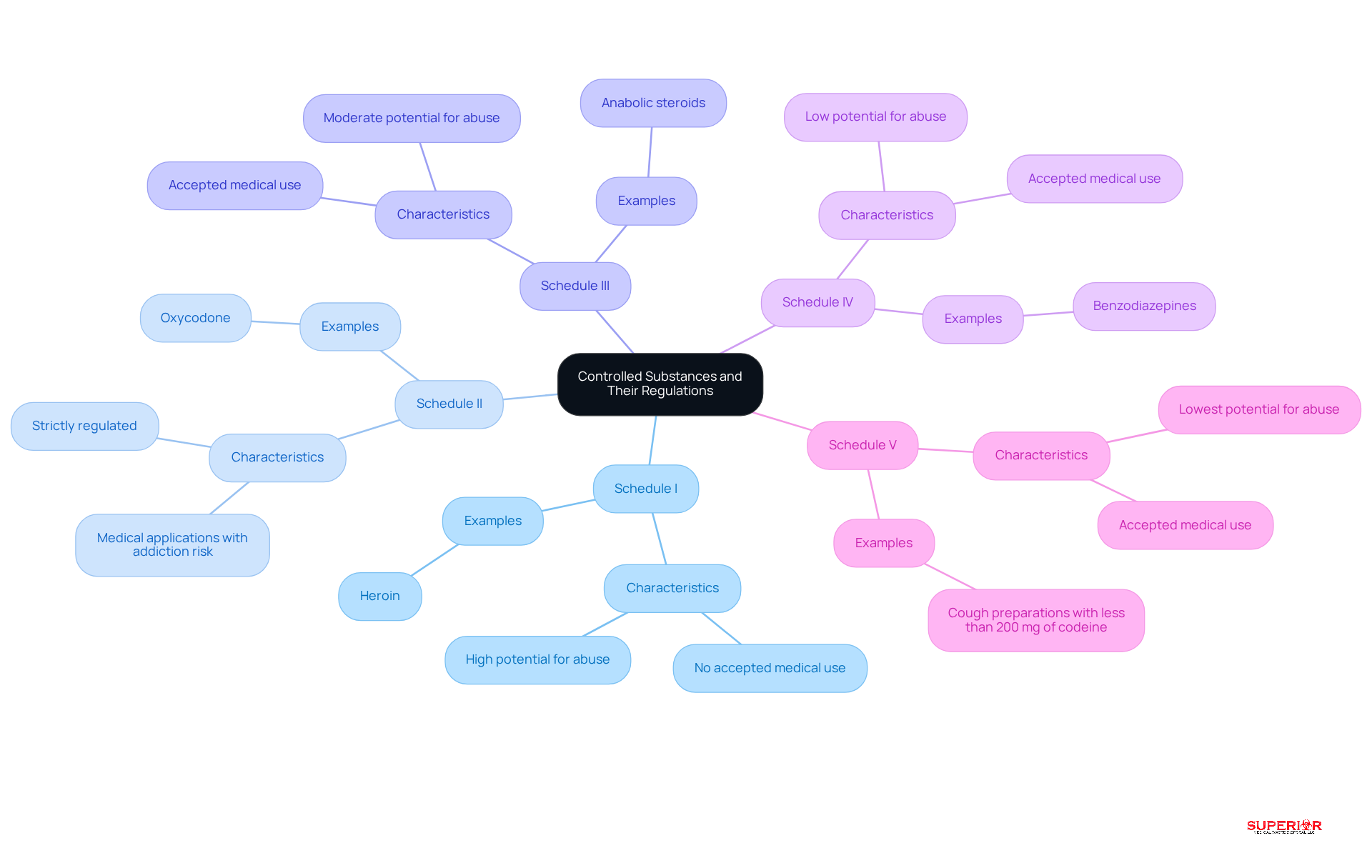
Controlled materials are classified into five schedules (I-V) based on their potential for abuse and approved medical applications. Schedule I drugs, such as heroin, are deemed to have no accepted medical use and a high potential for abuse. In contrast, Schedule II drugs, like oxycodone, are strictly regulated due to their medical applications and associated risk of addiction.
For healthcare professionals, understanding these classifications is vital for compliance with the Controlled Substances Act (CSA) and the regulations established by the Drug Enforcement Administration (DEA). Facilities must keep accurate records, documenting their acquisition, usage, and disposal of controlled substances. This diligence helps avoid penalties and ensures regulatory compliance.
Regular training is essential, as it keeps staff informed and compliant, fostering a culture of safety and accountability. Recent statistics indicate that compliance rates with the CSA in medical centers are concerning, highlighting the need for ongoing education and adherence to established practices.
As emphasized by DEA officials, maintaining accurate records is crucial for compliance and ensuring that controlled materials are managed responsibly.
Implement Safe Disposal Procedures for Controlled Substances
To implement safe disposal procedures, healthcare facilities must establish a clear protocol that includes several essential steps.
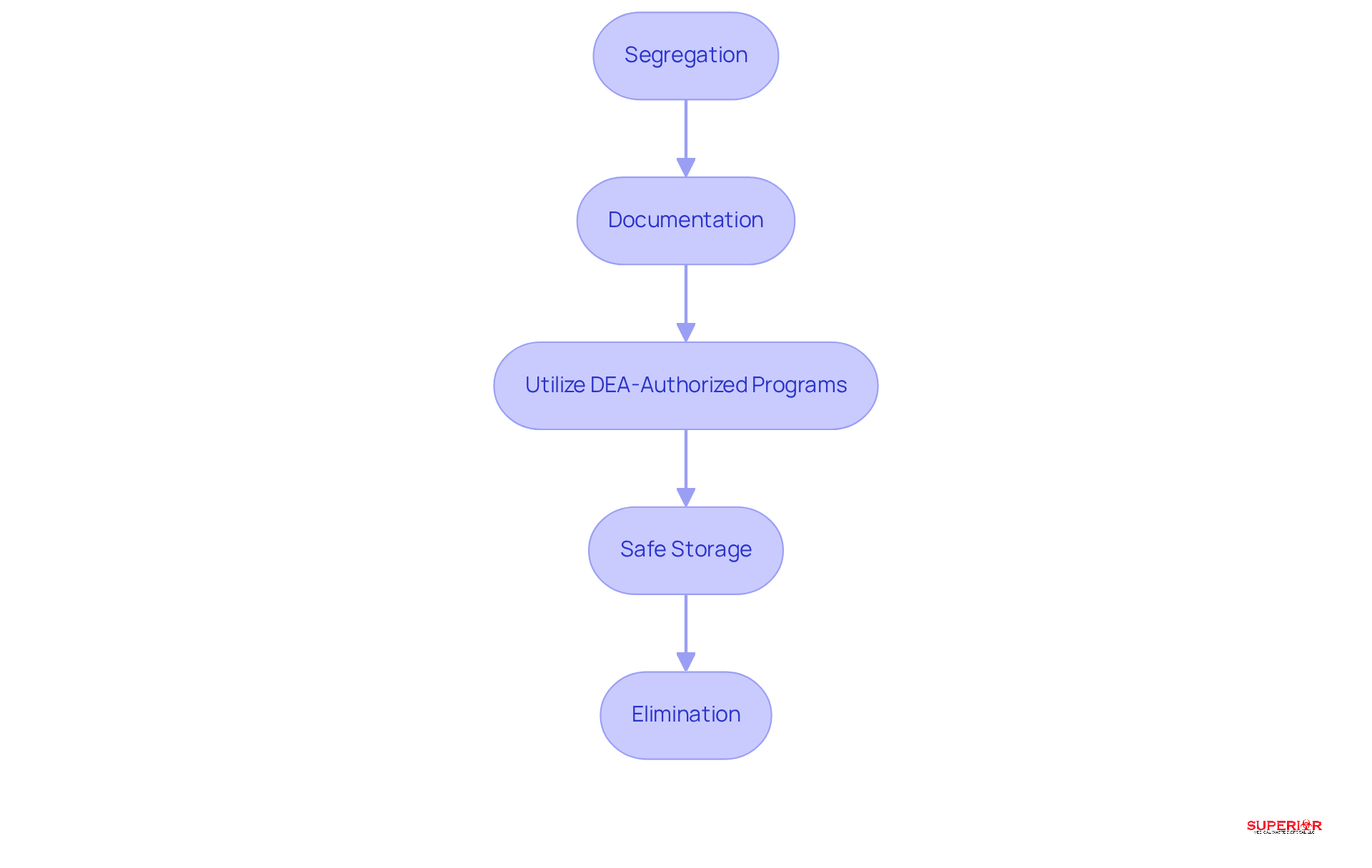
- Segregation: It is crucial to promptly separate regulated materials from other waste streams to prevent cross-contamination. This practice is vital for maintaining safety.
- Documentation: Comprehensive records of all regulated materials intended for elimination should be maintained, including quantities and methods of removal. This ensures accountability and traceability.
- Utilize DEA-Authorized Programs: Healthcare facilities should participate in take-back or mail-back alternatives for the elimination of unused medications. As of January 2026, these programs have been broadened to improve accessibility for both patients and medical providers.
- Safe Storage: Regulated materials awaiting removal must be kept in a locked, secure space to prevent unauthorized access. Recent data indicates that a significant number of medical establishments have adopted best practices. In Michigan, healthcare facilities have up to 90 days to store pharmaceutical waste before disposal, although this duration may vary by state.
- Elimination: Adhering to approved methods, such as incineration, is essential. This process is governed by the Environmental Protection Agency (EPA) and is utilized by authorized waste management sites to ensure that substances are rendered non-retrievable and comply with environmental regulations. Regular audits of disposal practices should be conducted to ensure compliance and identify areas for improvement, reinforcing the importance of these procedures in safeguarding public health.
The organization’s extensive network of partnerships with prominent organizations in the healthcare sector enhances their expertise and commitment to safety, ensuring that healthcare providers can trust their services.
Navigate Legal Guidelines for Controlled Substance Disposal
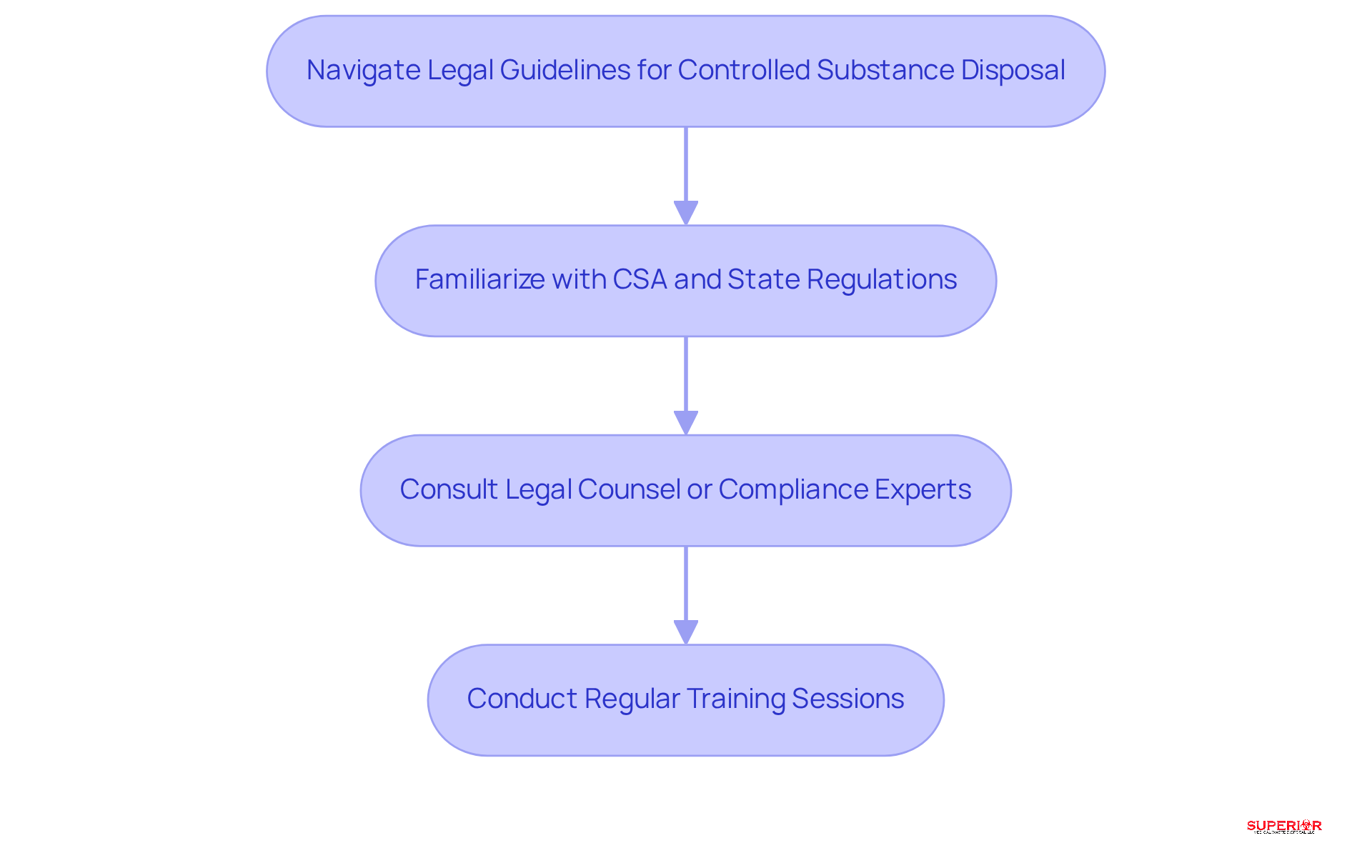
Facilities face a complex array of legal guidelines when it comes to the disposal of controlled materials. The Controlled Substances Act is a key regulation that mandates the proper disposal of controlled substances to prevent diversion and minimize environmental harm. It is crucial for facilities to familiarize themselves with state regulations, as these can differ significantly. For instance, certain states may impose additional requirements for the removal of hazardous waste.
To ensure compliance, it is advisable for facilities to consult with legal experts. Regular training sessions should also be conducted to keep staff informed about any changes in legislation, thereby reinforcing best practices in disposal procedures.
Recognize Risks of Improper Disposal of Controlled Substances
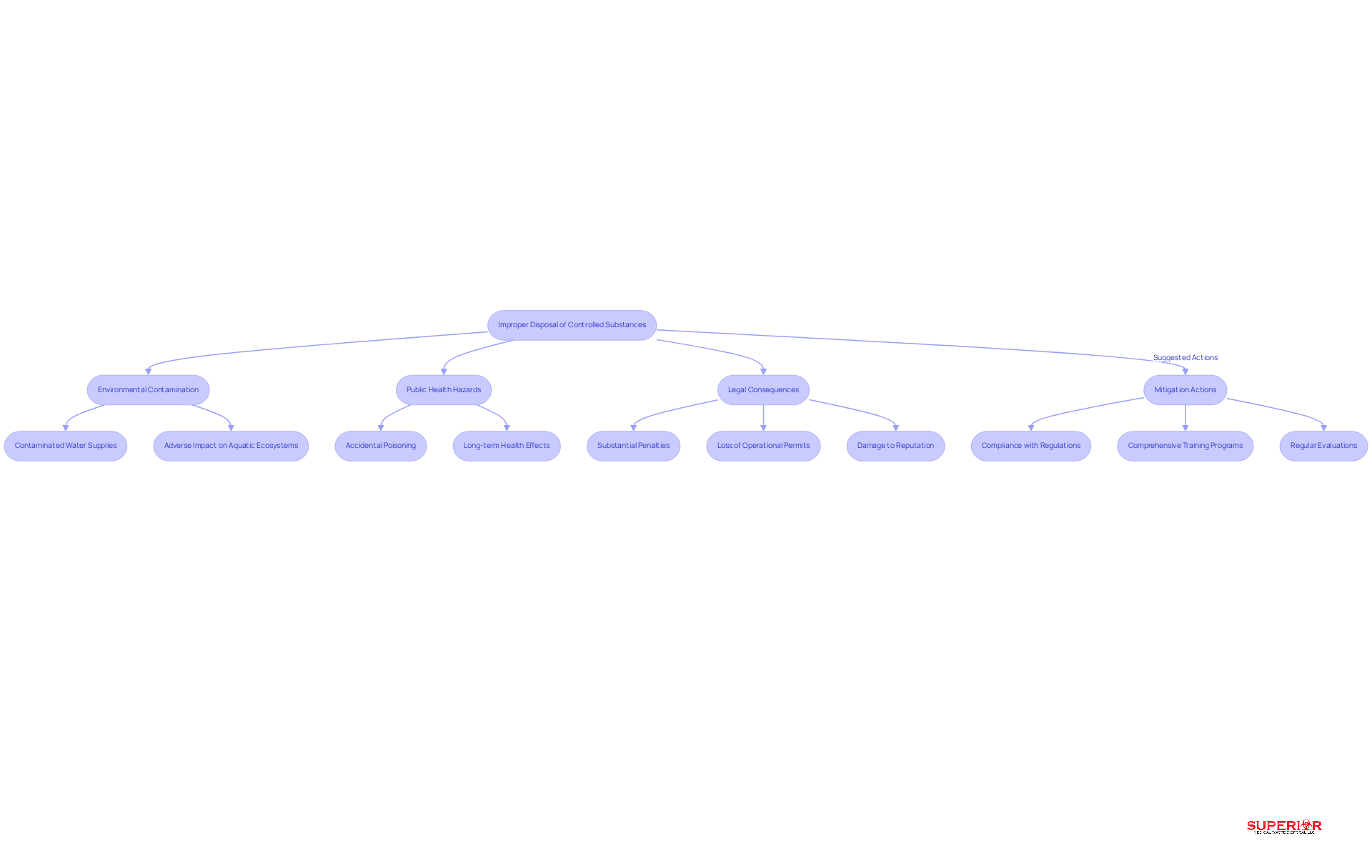
The improper disposal of controlled substances poses significant risks, including health hazards, environmental damage, and legal consequences. For example, flushing medications can contaminate water supplies, adversely impacting aquatic ecosystems and potentially entering the human food chain. Research indicates that trace pharmaceuticals are detectable in various water bodies, raising concerns about long-term health effects.
Furthermore, medical establishments that fail to adhere to regulations on the disposal of controlled substances may face fines, loss of operational permits, and damage to their reputation. To mitigate these risks, it is essential for healthcare facilities to train and implement safe disposal procedures for their staff.
Regular evaluations of the disposal practices can identify weaknesses and ensure alignment with established standards, ultimately protecting both public health and the environment.
Conclusion
The safe disposal of controlled substances in healthcare is not just a regulatory obligation; it is a vital aspect of public health and environmental safety. By following established best practices, healthcare facilities can manage these substances effectively, reducing the risks linked to improper disposal and ensuring compliance with the Controlled Substances Act and other relevant regulations.
Key insights from this article highlight the necessity of:
- Understanding the classifications of controlled substances
- Implementing stringent disposal procedures
- Navigating the intricate legal landscape surrounding these materials
Facilities must prioritize:
- Segregation
- Thorough documentation
- Participation in DEA-authorized programs
to guarantee that controlled substances are disposed of safely and responsibly. Regular training and compliance audits further reinforce the commitment to best practices, protecting both the community and the environment.
Neglecting proper disposal practices has far-reaching implications, impacting not only healthcare providers but also the wider community. By cultivating a culture of compliance and safety, healthcare facilities can mitigate the risks of environmental contamination and legal repercussions, paving the way for a healthier future. Embracing these best practices is essential for all stakeholders in the healthcare sector, ensuring that the management of controlled substances is conducted with the highest level of care and responsibility.
Frequently Asked Questions
What are controlled substances and how are they classified?
Controlled substances are materials classified into five schedules (I-V) based on their potential for abuse and approved medical applications.
What is the difference between Schedule I and Schedule II drugs?
Schedule I drugs, such as heroin, have no accepted medical use and a high potential for abuse. Schedule II drugs, like oxycodone, are strictly regulated due to their medical applications and associated risk of addiction.
Why is it important for medical institutions to understand controlled substance classifications?
Understanding these classifications is vital for compliance with the Controlled Substances Act (CSA) and the regulations established by the Drug Enforcement Administration (DEA), which helps avoid penalties and ensures patient safety.
What record-keeping practices are required for controlled substances?
Facilities must keep meticulous records of all controlled substances, documenting their acquisition, usage, and disposal.
Why is regular training on controlled substance regulations important?
Regular training keeps staff informed and compliant with regulations, fostering a culture of safety and accountability in medical institutions.
What do recent statistics indicate about compliance rates with the CSA in medical centers?
Recent statistics indicate that compliance rates with the CSA in medical centers are concerning, highlighting the need for ongoing education and adherence to established practices.
What is the overall significance of maintaining compliance with the CSA?
Maintaining compliance with the CSA is crucial for safeguarding public health and ensuring that controlled materials are managed responsibly.
List of Sources
- Understand Controlled Substances and Their Regulations
- What’s Your AI Strategy for 2026? The Roadmap to Future-Proofing AI Innovation – RapidScale (https://ncbi.nlm.nih.gov/books/NBK557426)
- drugabusestatistics.org (https://drugabusestatistics.org)
- HHS & DEA Extend Telemedicine Flexibilities for Prescribing Controlled Medications Through 2026 (https://hhs.gov/press-room/dea-telemedicine-extension-2026.html)
- mcdermottplus.com (https://mcdermottplus.com/insights/dea-extends-telemedicine-flexibilities-for-controlled-substance-prescribing-for-2026)
- Implement Safe Disposal Procedures for Controlled Substances
- Pharmacies Can Collect Unused Prescription Drugs Under New DEA Regulation | Pharmacy Times (https://pharmacytimes.com/view/pharmacies-can-collect-unused-prescription-drugs-under-new-dea-regulation)
- Best Practices for Disposing of Expired Controlled Substances (https://actenviro.com/best-practices-for-disposing-of-expired-controlled-substances)
- DPH Asks Residents to Use ‘Drug Take Back Day’ Events to Safely Dispose of Unused Prescription Medicines | South Carolina Department of Public Health (https://dph.sc.gov/news/dph-asks-residents-use-drug-take-back-day-events-safely-dispose-unused-prescription-medicines)
- Communities Nationwide Join DEA and its Partners to Safely Dispose of 571,000 pounds of Unwanted Medications (https://dea.gov/press-releases/2025/11/03/communities-nationwide-join-dea-and-its-partners-safely-dispose-571000-1)
- Nearly 630,000 Pounds of Unneeded Medications Collected on DEA’s National Prescription Drug Take Back Day (https://dea.gov/press-releases/2024/11/15/nearly-630000-pounds-unneeded-medications-collected-deas-national)
- Recognize Risks of Improper Disposal of Controlled Substances
- Pittsburg partners with DEA to promote safe drug disposal (https://fourstateshomepage.com/news/local/pittsburg-partners-with-dea-to-promote-safe-drug-disposal)
- Best Practices for Disposing of Expired Controlled Substances (https://actenviro.com/best-practices-for-disposing-of-expired-controlled-substances)
- Industry News (https://pharmecology.com/News/Recent)
- Group Urges FDA to Change Pharmaceutical Waste Policy from ‘Flush’ to Stewardship (https://waste360.com/medical-waste/group-urges-fda-to-change-pharmaceutical-waste-policy-from-flush-to-stewardship)
- DEA’s National Take Back Day Returns April 26 to Help Prevent Prescription Drug Misuse (https://dea.gov/press-releases/2025/04/22/deas-national-take-back-day-returns-april-26-help-prevent-prescription)
