Introduction
Selecting the appropriate pharmaceutical container is not merely a matter of convenience; it is a crucial factor that directly influences drug efficacy and patient safety. With a wide range of container types available, including vials and blister packs, it is essential for healthcare facilities to understand their specific uses and regulatory requirements. As the landscape of pharmaceutical regulations continues to evolve, the challenge for healthcare administrators is to maintain compliance while optimizing container management practices. How can they ensure that they are not only meeting safety standards but also enhancing operational efficiency in this dynamic environment?
Identify Types of Pharmaceutical Containers and Their Uses
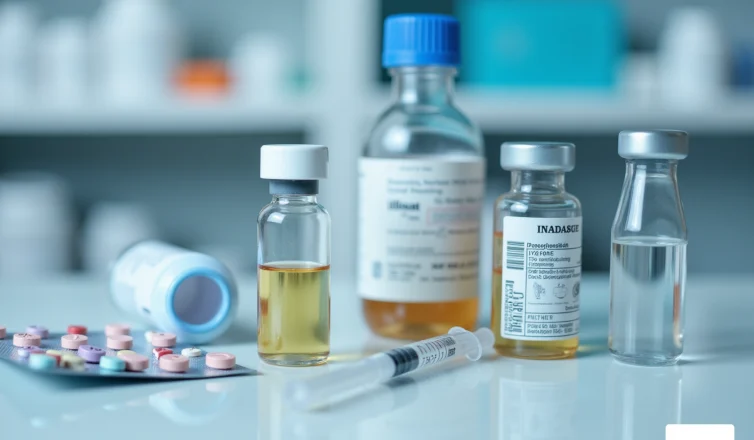
are available in various forms, each tailored to meet specific needs. The primary types include:
- Vials: These are utilized for liquids and powders, often sealed with rubber stoppers to maintain sterility.
- Blister Packs: Ideal for tablets and capsules, blister packs protect medications from moisture and contamination.
- Syringes: Designed for injectable medications, with the drug’s formulation to prevent adverse reactions.
- Bottles: Commonly used for both liquid and solid medications, bottles can be constructed from glass or plastic, depending on the stability requirements of the drug.
- Ampoules: These sealed glass vessels provide a sterile environment for sensitive medications.
Each type of receptacle serves a distinct purpose, making the selection of the container critical for safety and ensuring compliance.
Evaluate Regulatory Compliance and Safety Standards
When selecting a pharmaceutical container, assessing its compliance is crucial. This ensures that the packaging meets necessary safety and quality standards, ultimately protecting patient health.
It is vital to confirm that containers adhere to the standards established by the FDA. This includes ensuring safety and meeting labeling requirements. Good Manufacturing Practices (GMP) require that containers be produced in accordance with regulations. This practice guarantees that the packaging does not compromise the drug’s quality. For specific medications, incorporating child-resistant features is mandatory to prevent accidental ingestion by children. The material of the container must be compatible with the drug. Any reaction between the vessel material and the medication could alter its efficacy or safety.
Regulatory agencies can help facilities maintain these standards, thereby ensuring compliance and avoiding costly fines.
Implement Effective Management Practices for Container Use
To optimize the use of pharmaceutical containers, healthcare facilities should adopt several key management practices:
- Inventory Management: Regular monitoring of container inventory is crucial. This practice ensures adequate supplies while promptly removing expired or damaged containers from circulation. Such proactive measures help prevent compliance issues and guarantee that only safe, usable vessels are available.
- Training staff on the proper handling and storage of containers through comprehensive programs is essential. Utilizing resources, such as those offered by Superior Medical Waste Disposal, can significantly enhance staff knowledge on compliance-related topics. This minimizes the risk of contamination or damage. Facilities that prioritize training have reported improvements, with statistics indicating a decrease in incidents related to improper handling. For example, labor for handling and sharps was reduced by nearly 50% at Rome Hospital, highlighting the effectiveness of robust training programs.
- Clearly labeling all containers with pertinent information, such as contents, expiration dates, and handling instructions, is vital. This practice not only prevents misuse but also facilitates swift identification and adherence to regulatory standards.
- Proper disposal ensures that pharmaceutical waste is disposed of correctly. This approach reduces environmental impact and aligns with optimal strategies for handling waste from containers.
Healthcare administrators must also recognize that penalties for non-compliance with regulations can be severe. Federal EPA violations can incur penalties of up to $75,000 per day per violation. By implementing these protocols, healthcare facilities can enhance safety, ensure regulatory adherence, and improve overall operational efficiency.
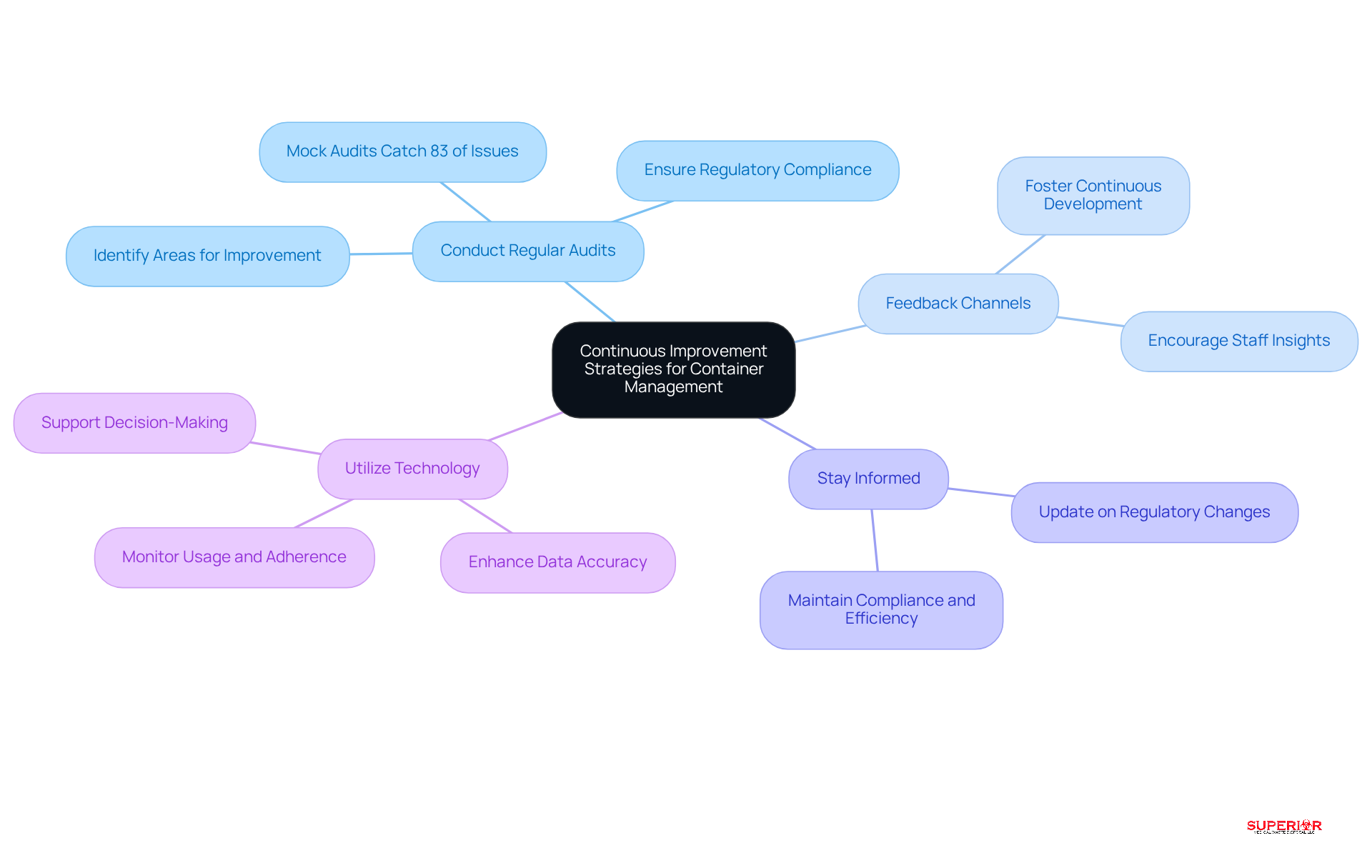
Adopt Continuous Improvement Strategies for Container Management
To ensure ongoing compliance and efficiency in pharmaceutical management, healthcare facilities should adopt several key strategies:
- Conduct Regular Audits: Establish a schedule for periodic audits of container practices. These audits are crucial for identifying areas that require improvement and ensuring adherence to regulatory standards. Research indicates that audits can uncover up to 83% of regulatory issues before they are flagged by authorities, significantly reducing the risk of penalties, which can average $5.3 million for regulatory violations.
- Feedback Channels: Develop robust avenues for feedback on handling procedures. This fosters a culture of continuous development, allowing teams to share insights and suggest enhancements that can lead to better adherence and compliance.
- Stay Informed: Regularly update your understanding of changes in regulations and industry standards. Staying informed ensures that storage management practices remain compliant and efficient, which is vital in an industry where the cost of non-compliance can reach $5.3 million.
- Utilize Technology: Implement technology for container management. Technology can streamline processes, enhance data accuracy, and offer real-time insights into adherence status, ultimately supporting improved decision-making.
By adopting these strategies, healthcare facilities can significantly enhance their management practices for pharmaceutical containers, ensuring safety and compliance while optimizing efficiency.
Conclusion
Selecting and managing pharmaceutical containers is crucial for ensuring medication safety and efficacy. The right container choice not only preserves drug integrity but also adheres to regulatory standards that safeguard patient health. A solid understanding of the various container types, their specific applications, and the importance of regulatory compliance is essential for effective pharmaceutical container management.
This article has explored key practices for selecting and managing pharmaceutical containers. From identifying appropriate container types – such as vials, blister packs, and syringes – to ensuring compliance with FDA regulations and Good Manufacturing Practices, each element is vital for maintaining medication quality. Furthermore, effective management practices, including inventory monitoring, staff training, and the implementation of continuous improvement strategies, are critical for sustaining operational efficiency and compliance.
The significance of selecting and managing pharmaceutical containers cannot be overstated. By prioritizing best practices and staying informed about regulatory changes, healthcare facilities can enhance patient safety and mitigate the risk of costly penalties. Embracing these strategies fosters a culture of compliance and positions organizations for long-term success in the dynamic pharmaceutical landscape.
Frequently Asked Questions
What are the main types of pharmaceutical containers?
The main types of pharmaceutical containers include vials, blister packs, syringes, bottles, and ampoules.
What is the purpose of vials in pharmaceutical packaging?
Vials are utilized for liquids and powders, often sealed with rubber stoppers to maintain sterility.
How do blister packs protect medications?
Blister packs are ideal for tablets and capsules as they protect medications from moisture and contamination.
What are syringes used for in pharmaceuticals?
Syringes are designed for injectable medications and must be compatible with the drug’s formulation to prevent adverse reactions.
What materials are commonly used for pharmaceutical bottles?
Pharmaceutical bottles can be constructed from glass or plastic, depending on the stability requirements of the drug.
What is the function of ampoules in pharmaceutical packaging?
Ampoules are sealed glass vessels that provide a sterile environment for sensitive medications.
Why is it important to select the appropriate pharmaceutical container?
Selecting the appropriate container is crucial for preserving drug integrity and ensuring patient safety.
List of Sources
- Evaluate Regulatory Compliance and Safety Standards
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- Current Good Manufacturing Practice (CGMP) Regulations (https://fda.gov/drugs/pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations)
- Regulatory Compliance for Pharmaceutical Industry | 2026 Guide (https://compliancequest.com/bloglet/regulatory-compliance-for-pharmaceutical-industry)
- Contract Manufacturing Oversight: 2026 FDA Enforcement Data | IntuitionLabs (https://intuitionlabs.ai/articles/contract-manufacturing-oversight-fda-enforcement-2026)
- Implement Effective Management Practices for Container Use
- The top inspirational quotes from famous healthcare personalities (https://devorerecruiting.com/blog/top-inspirational-quotes-from-famous-healthcare-personalities.htm)
- 2026 Medical Waste Compliance Guide: 2025 Reg Changes & Beyond (https://sdmedwaste.com/post/healthcare-waste-regulations-compliance-guide)
- Case Studies | Daniels Health (https://danielshealth.com/case-studies)
- Top 100 Inspirational Workplace Safety Quotes (https://damotech.com/blog/inspirational-quotes-warehouse-safety)
- 30 Inspirational Quotes for Healthcare Workers (https://chicomm.com/blog/30-inspirational-quotes-for-healthcare-workers)
- Adopt Continuous Improvement Strategies for Container Management
- Regulation first: Compliance is the foundation of pharma packaging (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/pharmaceutical-packaging-news/regulation-first-compliance-is-the-foundation-of-pharma-pack)
- Pharma Fines Exposed: How Much Non-Compliance REALLY Costs – Pharmacy Business Hub (https://pharmabusinesshub.com/pharma-fines-exposed-how-much-non-compliance-really-costs)
- niagarainstitute.com (https://niagarainstitute.com/blog/quotes-improving-processes)
- Inspiring Quotes for Continuous Improvement (https://kpifire.com/blog/quotes-about-continuous-improvement)
- osfinnovation.org (https://osfinnovation.org/about-osf-innovation/case-studies/continuous-improvement-lean)